Impact of Core Fucosylation on Antibody Effector Functions using TransGLYCIT®
Application
Functional comparison of antibody glycoforms by combining glycoengineering with ADCC and ADCP reporter assays to assess Fc effector activity.

Antibody-dependent cellular cytotoxicity (ADCC) and antibody-dependent cellular phagocytosis (ADCP) effector functions represent the mode-of-action of many therapeutic antibodies. ADCC and ADCP rely on the binding of the antibody Fc domain to specific Fc-gamma receptors on the effector cells which activate a number of downstream pathways.
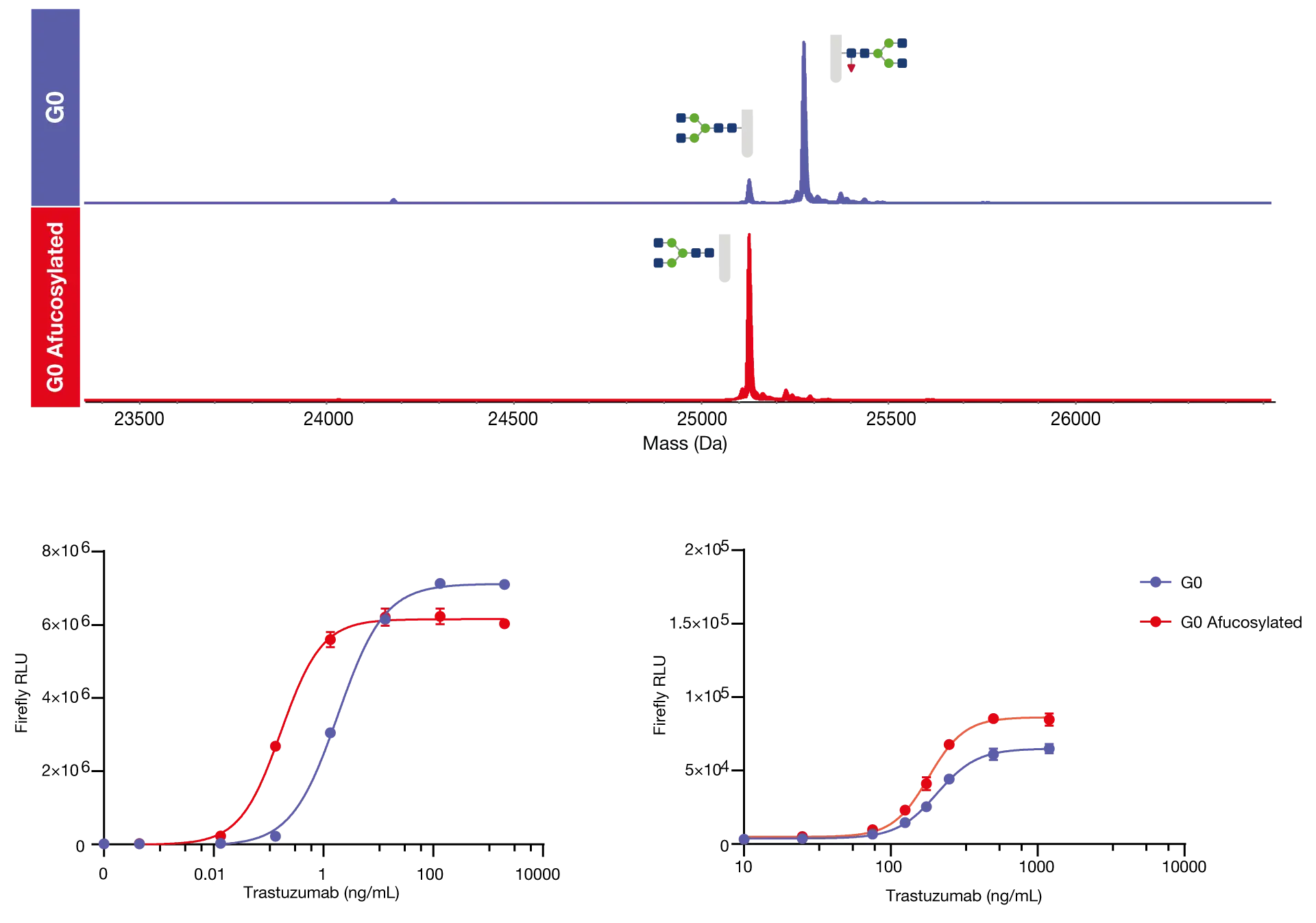
In collaboration with Svar Life Science, we generated glycoengineered trastuzumab containing homogeneous G0F glycans and core afucosylated G0 glycans using TransGLYCIT. This allows a direct comparison between the performance of the fucosylated and afucosylated glycoforms in reporter gene bioassays where specific target and effector cells were used to determine the ADCC and the ADCP of the modified antibodies. Following FabRICATOR digestion, the deconvoluted mass spectra of the glycoengineered trastuzumab scFc domain demonstrates the highly efficient generation of the G0 and G0F glycoforms using TransGLYCIT.
Efficient generation of glycoengineered antibodies with homogeneous glycan profiles
ADCC and ADCP functional assays (iLite®, SVAR) were carried out using the glycoengineered trastuzumab to determine the impact of core fucosylation on antibody effector function. In the ADCC reporter assay, we can see a clear shift to the left in the afucosylated antibody which indicates a clear and considerable increase in the ADCC potency, and in this instance, there was a calculated 10-fold decrease in EC50 following core afucosylation. In the ADCP assay we observe that there was not significant horizontal shift, which indicates that ADCP potency is similar between the two samples, however, the clear vertical shift that we observe following afucosylation indicates a considerable increase in ADCP efficacy
Core afucosylation increases ADCC potency and ADCP efficacy
The use of TransGLYCIT to remodel antibody glycan profiles allows a direct functional assessment of specific glycoforms to be performed using bioassays of this type which can support product development and increase functional understanding.
Determination of relationship between Fc N-glycan structure and antibody effector function
Related Products
TransGLYCIT® Remodeling Afucosylated
Glycan remodeling of human IgG with the G0, G1, G2, G2S2 or Man5 glycoform, with core afucosylation.

Resources
TransGLYCIT®
Download Product Folder

TransGLYCIT® to Evaluate Fc Effector Function of Therapeutic Antibodies
Download Scientific Poster

TransGLYCIT® for Fc-specific N-glycan Remodeling of Human IgG
Download Scientific Poster






