SmartEnzymes™ for Gene Therapy
Download Product Folder


SmartEnzymes™
Xork Preclinical Grade is a next-generation IgG-specific cysteine protease from a non-human pathogen, with validated low endotoxin levels, developed for in vivo preclinical research into IgG-mediated disease and immunomodulation. It digests human IgG at a single site below the hinge, generating homogeneous F(ab’)2 and Fc fragments for rapid reduction of pathogenic or neutralizing IgG – with minimal pre-existing anti-enzyme antibodies enabling repeat-dose studies in gene therapy, autoimmunity, and transplantation.
No need for reducing agents or co-factors
Preclinical grade format of the Xork enzyme for digestion of IgG. This formulation has low levels of endotoxins (<0.1 EU / vial).
Contact Us
Preclinical grade format of the Xork Fc enzyme for digestion of IgG, an Fc-domain variant of Xork. This formulation has low levels of endotoxins (<1 EU / vial).
Contact Us
Xork Preclinical Grade is a cysteine protease that digests human IgG at a single site below the hinge, generating a homogeneous pool of F(ab’)2 and scFc fragments. Like FabRICATOR, Xork combines strong IgG-digesting activity with high substrate specificity, which makes it suitable for use in complex samples and in vivo.
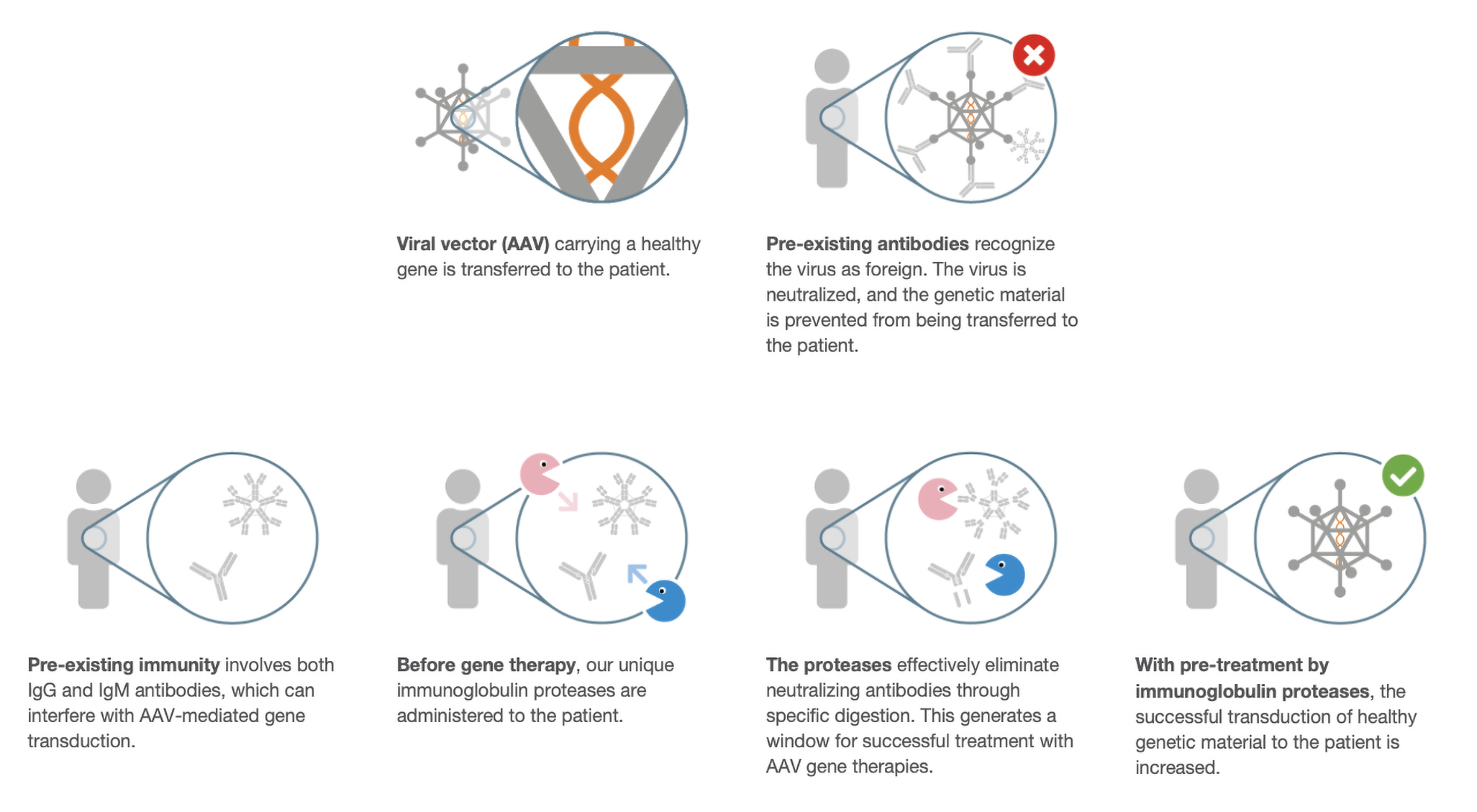
Xork is derived from a non-human pathogen, so pre-existing anti-enzyme antibodies are not expected in the general human population – a key differentiator from streptococcal enzymes such as IdeS, where widespread prior exposure can reduce activity and limit repeat dosing. This expands the scope of in vivo immunomodulation research, supporting cyclical regimens in autoimmunity, repeated donor-specific antibody removal in transplantation, and strategies to overcome anti-AAV neutralizing antibodies in gene therapy.
Xork Preclinical Grade is recombinantly produced, highly purified, and manufactured with validated and controlled low endotoxin levels, making it suitable for a wide range of in vivo preclinical studies. An Fc-domain variant, Xork Fc, is also available.
A method to detect the Xork enzyme is available. Learn more about Anti-Xork, an affinity-purified antibody targeting the Xork enzyme.
Efficient IgG cleavage with a low immunogenicity risk enzyme, supporting clearance of neutralizing antibodies to improve gene therapy administration.
Download Scientific Poster

All rights reserved. Genovis products may be covered by one or more patents, trademarks and copyrights owned by Genovis AB or licensed from third parties. For more information about commercial rights, please contact the Genovis team at licensing@genovis.com.
Genovis products are intended for research use only. They are not intended to be used for therapeutic or diagnostic purposes in humans or animals. All goods and services are sold subject to Genovis’ General Terms and Conditions of Sale.
© Genovis AB