Efficient Desialylation of Complex Glycoproteins using SialEXO™
Performance Activity
Complete desialylation of complex glycoproteins to simplify glycan profiles and enable more confident protein characterization.

Sialylation is a major source of heterogeneity in highly complex glycoproteins, where terminal sialic acids can strongly influence charge, and add significant analytical complexity. Complete removal of sialic acid can simplify glycan profiles and reduce masking effects, enabling clearer access to characterize the underlying protein structure and remaining glycan features with greater confidence.
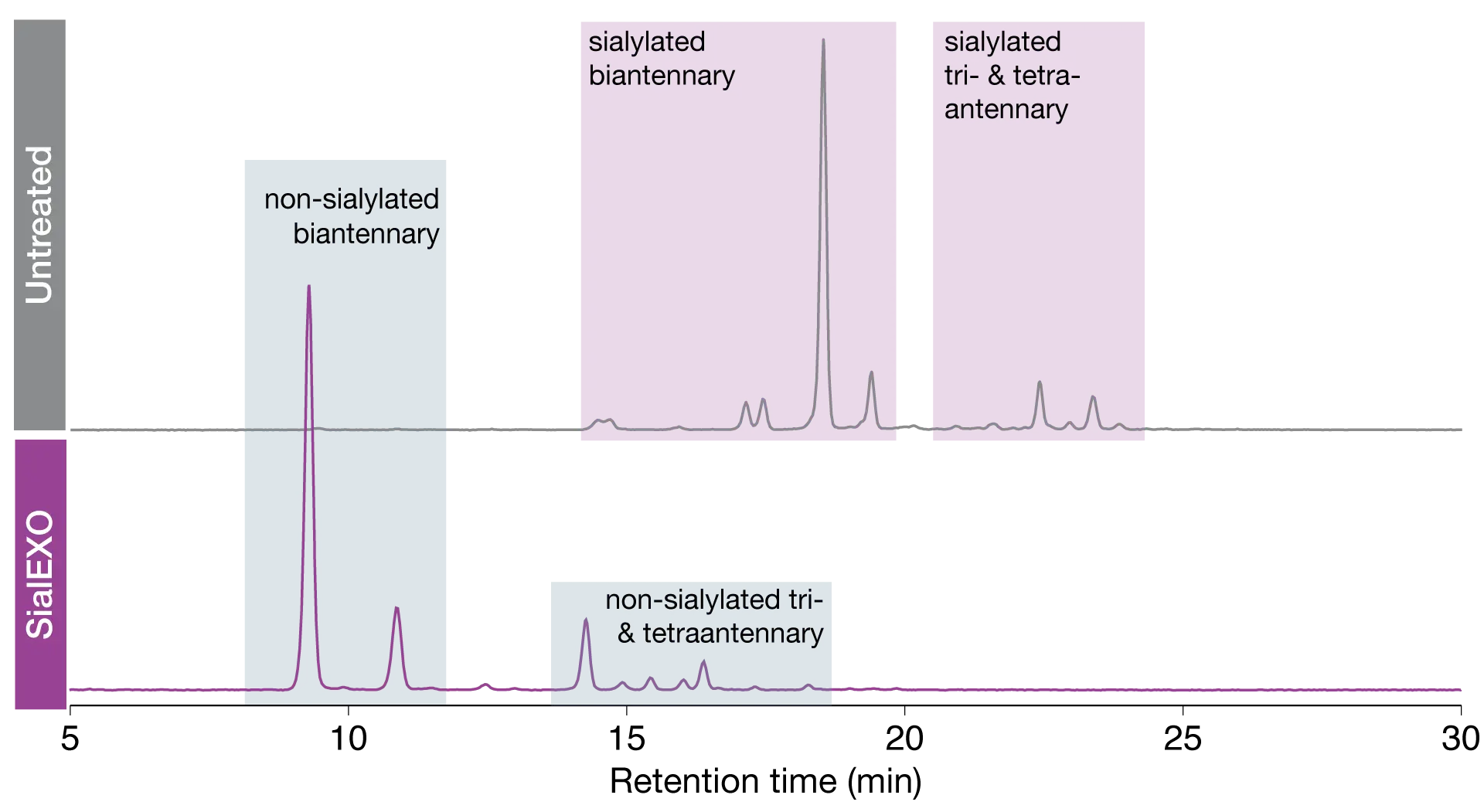
Here, we demonstrate the efficiency of sialic acid hydrolysis from a highly complex protein substrate. In this example, we are using human C1-inhibitor which is a challenging substrate containing 6 sites of N-linked glycosylation and high levels of sialylation. We performed released N-glycan analysis on human C1-inhibitor samples with and without SialEXO incubation. The released glycan profile of the native protein contains predominantly bi-tri and tetra-antennary sialylated glycans species. Following incubation with SialEXO, we can observe the sialic acids have been efficiently removed and the glycan profile now shows completely desialylated glycan structures with no sialic acid containing glycans remaining. This highlights the high efficiency of SialEXO as a tool for complete desialylation of complex glycoprotein samples.
Efficient and complete desialylation of highly challenging substrates
Related Products
Resources
SialEXO™
Download Product Folder

SmartEnzymes™ for the Glycosylation Charcterization of Glycoproteins
Download Scientific Poster





