GlyCLICK®
Download Product Folder

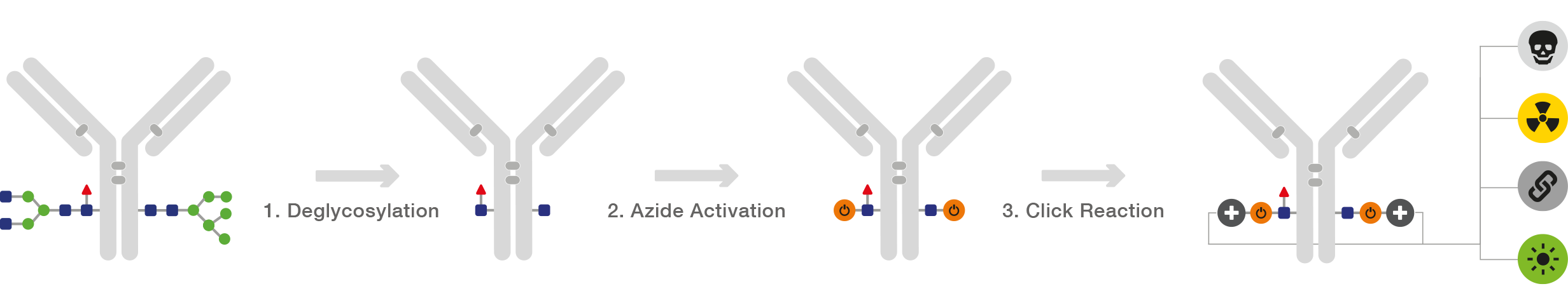
GlyCLICK is a three-step conjugation technology for site-specific and quantitative conjugation of IgG from several species and subclasses.

With GlyCLICK, specific Fc N-glycan hydrolysis allows for site-specific conjugation at the core GlcNAcs using robust click-chemistry and results in a degree of label (DOL) or drug-antibody ratio (DAR) of 2. The reliable performance ensures quantitative conjugation and intact immunoreactivity for sensitive applications.
The technology is available in a range of kit formats, including conjugation with fluorophores, biotin, DFO, or toxins for ADC development. The format “Azide Activation” renders the antibody azide-activated, for conjugation with any alkyne-reactive label of choice.
Human IgG1-4, IgG from mouse, rabbit, rat, monkey, sheep, goat, cow and horse
2-3 day protocol
Fluorophore, Biotin, DFO, MMAE, PNU, Azide Activation
Conjugation occurs at the Fc N-glycan sites
Site-specific conjugation of IgG with azide-alkyne click chemistry
Buy product




Site-specific ADC generation with defined DAR, preserving antigen binding and delivering potent, controlled cytotoxicity in target cells.
Improved PET/CT tumor targeting and biodistribution using site-specific antibody labeling with consistent DOL and preserved in vivo performance.
Supports high-quality immunofluorescence imaging with consistent signal intensity and reliable quantification through defined antibody labeling.
Higher-resolution flow cytometry with improved signal intensity and a 10-fold increase in separation index using site-specific antibody labeling.
Download Scientific Poster

Download Scientific Poster

Download Scientific Poster

Watch video
Download Scientific Poster

Download Scientific Poster

The GlyCLICK technology can be used to conjugate IgG with Desferrioxamine (DFO) using the GlyCLICK DFO kit. The DFO is a chelating agent for radiolabeling with the radioisotope Zirconium-89 (89Zr) for PET-imaging. The Azide Activation kit can be used to conjugate a chelator of choice if it is alkyne-modified (carrying for example DIBO, DBCO or BCN) to be compatible with the click chemistry.
Problems can arise from freezing of the conjugates. We cannot guarantee the quality after freezing and therefore recommend storage at +4°C.
GlyCLICK is available in kit formats with AlexaFluor®488, AlexaFluor®555, AlexaFluor®647 or in the Azide Activation kit format for conjugation of a label of choice. With the fluorescent label being alkyne-modified (carrying for example DIBO, DBCO or BCN) it is possible to combine it with the GlyCLICK azide activation kit.
All the steps (deglycosylation, azide activation and click reaction) in GlyCLICK are highly efficient. Normally, all molecules are conjugated when using GlyCLICK, resulting in DOL=2. On the application page we have shown by MS-analysis that all molecules have been conjugated and there is no remaining un-conjugated material.
Characterization using LC-MS will give a complete and clear picture of mass shift associated with successful conjugation if the antibody is analyzed reduced or fragmented. LC or LC-MS analysis of HC from a reduced sample or scFc fragments generated by FabRICATOR digestion provides distinct elution peaks for conjugated scFc or mass shifts corresponding to the degree of labelling (DOL) following conjugation. On the application page you can see an example of such analysis. Analysis of fluorescently labeled antibodies can also be performed by absorbance measurements and calculate the DOL using the Extinction coefficient for the antibody and the label and the correction factor for the label.
All rights reserved. Genovis products may be covered by one or more patents, trademarks and copyrights owned by Genovis AB or licensed from third parties. For more information about commercial rights, please contact the Genovis team at licensing@genovis.com.
Genovis products are intended for research use only. They are not intended to be used for therapeutic or diagnostic purposes in humans or animals. All goods and services are sold subject to Genovis’ General Terms and Conditions of Sale.
© Genovis AB