ImpaRATOR™ Lyophilized
Lyophilized enzyme for digestion of mucin-type O-glycoproteins and peptides, including sialylated O-glycan species

Application
Broad O-glycan substrate acceptance efficiently generating informative O-glycopeptides while retaining glycan structure information.
Etanercept is a fusion protein consisting of the extracellular domain of the TNF receptor linked to the Fc portion of human IgG1. The protein contains a cluster of up to 13 O-glycosylated Ser/Thr residues just above the Fc region, making it a very difficult biopharmaceutical to characterize structurally.
To demonstrate the broad glycan specificity of ImpaRATOR, three samples of etanercept with different glycan compositions were prepared:
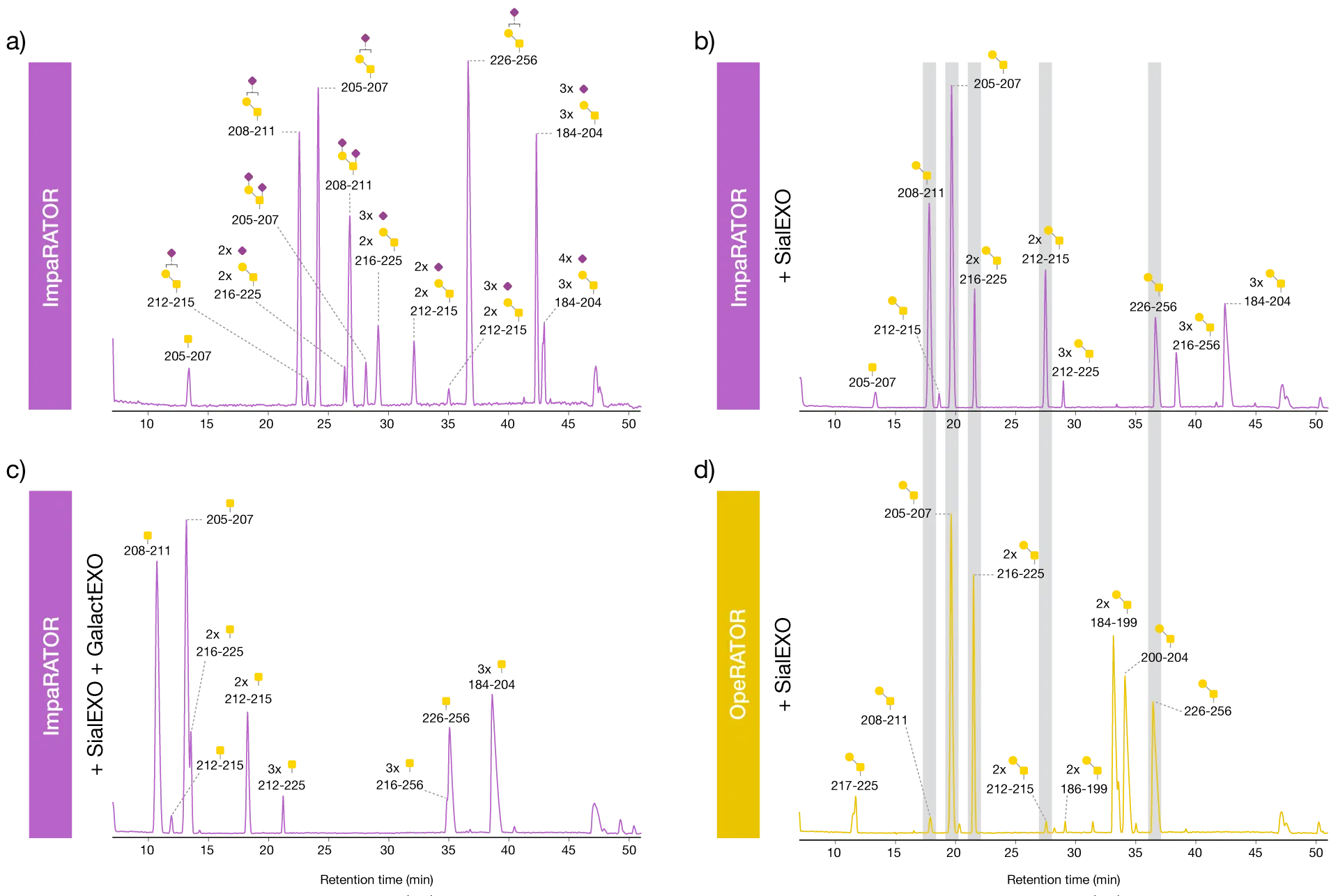
The three samples were treated with ImpaRATOR and FabRICATOR in a one-pot reaction (Fig. 1). As a comparison, SialEXO-treated etanercept was treated with OpeRATOR and FabRICATOR. The resulting peptides were reduced, alkylated and analyzed by HILIC-MS. FabRICATOR was included in the reaction to separate the Fc region from the most C-terminal glycopeptide to facilitate its characterization. Analysis of the ImpaRATOR-treated samples showed that ImpaRATOR digested all three substrates well and generated similar glycopeptides, but with the expected different glycan species (Fig. 2). The sample treated only with ImpaRATOR contained more variants than the other samples since each peptide was detected with several glycan structures.
ImpaRATOR digestion generates O-glycopeptides while retaining glycan structures

The observed glycan species were mainly mono- and di-sialylated core 1 structures, which is in accordance with the expected ones for etanercept. The SialEXO-pretreated samples digested with ImpaRATOR or OpeRATOR both resulted in peptides with naked core 1 structures, with the exception of peptide T205-P207 carrying the Tn antigen observed in the ImpaRATOR-digested sample. Etanercept pretreated with SialEXO and GalactEXO followed by digestion with ImpaRATOR resulted in peptides with only the Tn antigen. Together, this data demonstrates the high glycan substrate acceptance of ImpaRATOR.
ImpaRATOR recognizes a wide range of glycan substrates
The ImpaRATOR-treated etanercept displayed a slightly different digestion pattern than the corresponding desialylated OpeRATOR-treated sample. For example, OpeRATOR digested between two O-glycosylated amino acids more efficiently than ImpaRATOR (for details, see section below). Etanercept with intact sialic acids or the Tn antigen was only digested very weakly by OpeRATOR (data not shown).
Different digestion profiles for ImpaRATOR and OpeRATOR
Lyophilized enzyme for digestion of mucin-type O-glycoproteins and peptides, including sialylated O-glycan species

Download Scientific Poster

Download Product Folder

Download Product Folder
