Rapid Determination of Fc Oxidation using FabRICATOR® and GlycINATOR™
Application
Fast and reliable middle-level analysis of antibody oxidation using FabRICATOR and GlycINATOR, supporting robust QC of critical quality attributes.

Monitoring of critical quality attributes of therapeutic antibodies is necessary throughout the discovery, development, and commercial production of therapeutic antibodies. Parameters that require characterization and control include both fundamental information, such as the amino acids sequence of the protein and post-translational modifications such as glycosylation, as well as changes that may occur during storage and formulation, such as methionine oxidation. Traditionally, methionine oxidations on antibodies are studied using bottom-up approaches and require lengthy sample preparations with trypsin and result in complex data sets. A middle-level approach for antibody subunit oxidation determination has been developed by Sokolowska and colleagues1 and more recently validated2 for use as a batch release assay within Quality Control (QC) labs.
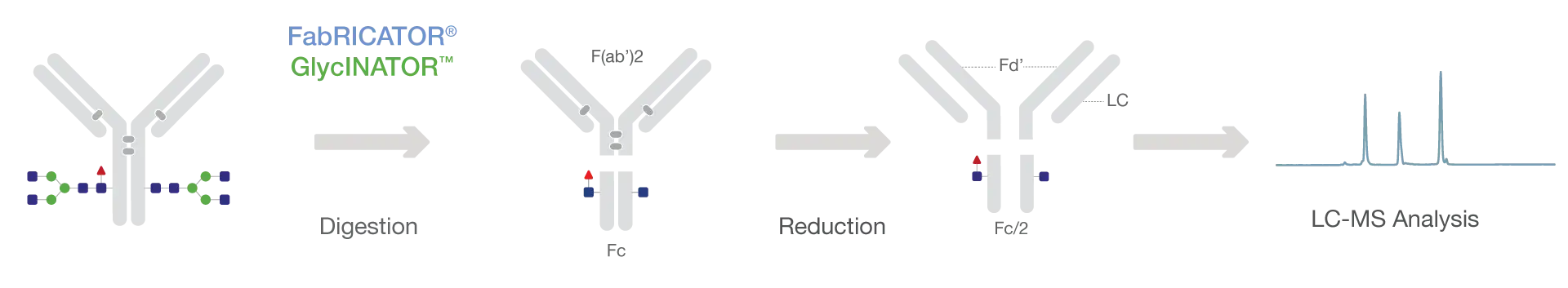
Here, we demonstrate a variation of the validated approach which combines FabRICATOR and GlycINATOR (Fig. 1). The specificity of these enzymes means that they can be combined in a single digestion for rapid and efficient sample preparation. GlycINATOR specifically removes the antibody Fc-glycans which significantly reduces the overall heterogeneity making it possible to identify Fc oxidation states. Following digestion and reduction, the samples can be analysed by LC-MS and automated data processing methods can be applied which are amenable to QC applications.
mAb oxidation analysis at the subunit level suitable for Quality Control applications
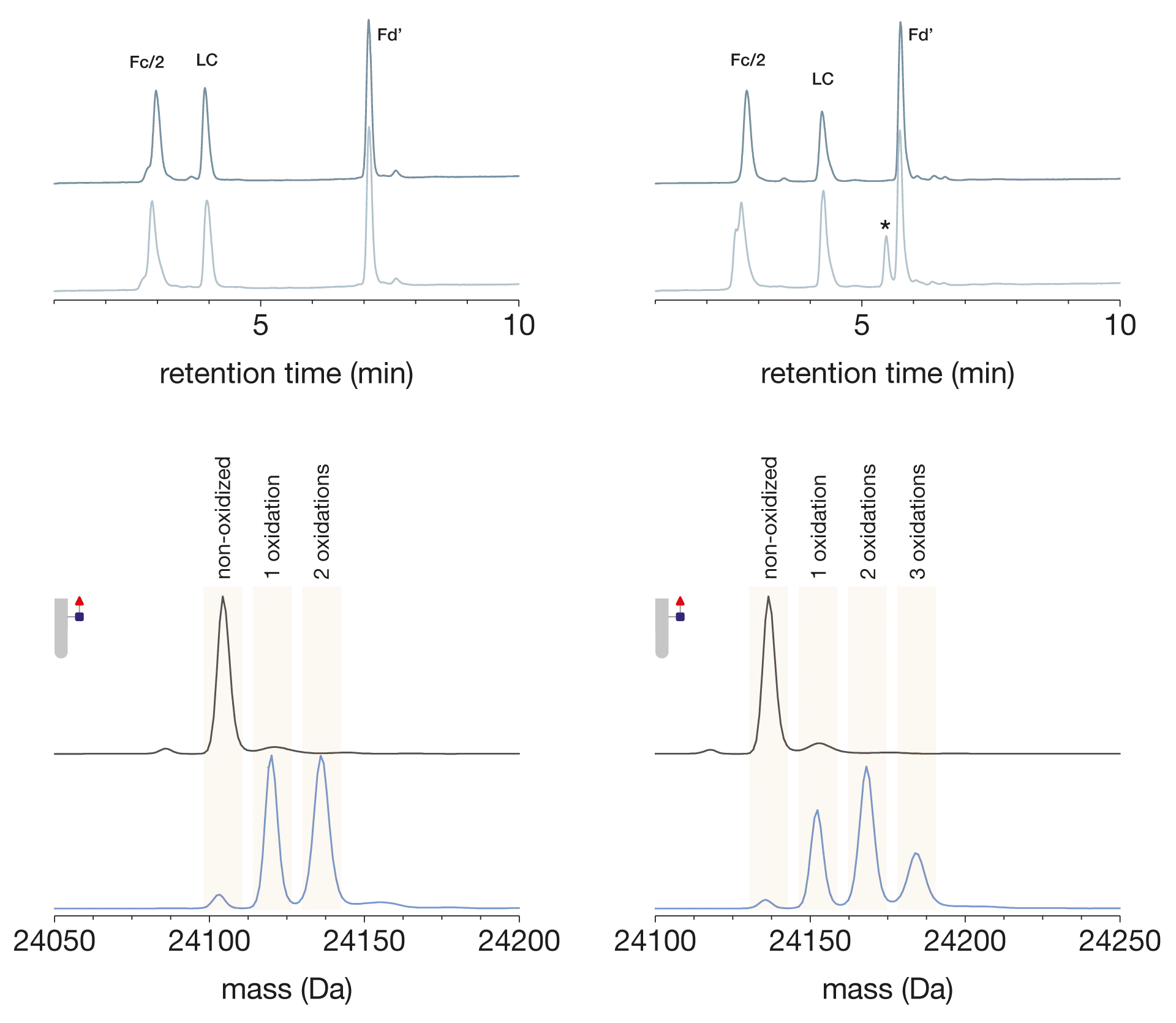
The characterization of two human IgG1 molecules, adalimumab and trastuzumab, is shown here (Fig. 2), however, we have also applied this approach to IgG2 and IgG4 molecules which highlights the capability to use this approach as a platform assay for many different mAbs. To confirm this method’s ability to effectively identify and quantify oxidized species, forced oxidation samples were analyzed following incubation with hydrogen peroxide.
Platform approach for fast and simple oxidation analysis at the middle-level
Following digestion with the combination of FabRICATOR and GlycINATOR, the oxidized species were well resolved at the mass spectral level. The relative amounts of non-oxidized species, as well as species containing 1, 2 or 3 oxidations, in the Fc subunit could be easily determined and quantified. In the case of trastuzumab, we could also identify additional oxidation within the Fd subunit’, highlighting the strength of a middle-level approach for critical quality attribute (CQA) characterization. This global oxidation approach is fast, simple, and is also compatible with automated sample preparation strategies which we have also demonstrated.
1.Sokolowska, I. et al., 2017. Subunit mass analysis for monitoring antibody oxidation. mAbs, 9(3), pp.498–505.
2. Sokolowska, I. et al., 2020. Implementation of a High-Resolution Liquid Chromatography-Mass Spectrometry Method in Quality Control Laboratories for Release and Stability Testing of a Commercial Antibody Product. Analytical Chemistry, 92(3), pp.2369–2373.
Fast and simple workflow which is automation compatible
Related Products
Resources
AppNote SmartEnzymes™ for mAb Oxidation
Download Application Notes

How FabRICATOR® works
Watch video



