FabRICATOR® Xtra LALA Lyophilized
Lyophilized enzyme for below hinge digestion of hinge-mutated IgG, including LALA

Efficient digestion of hinge-mutated antibodies, generating fragments for comprehensive subunit LC-MS analysis of engineered IgG formats

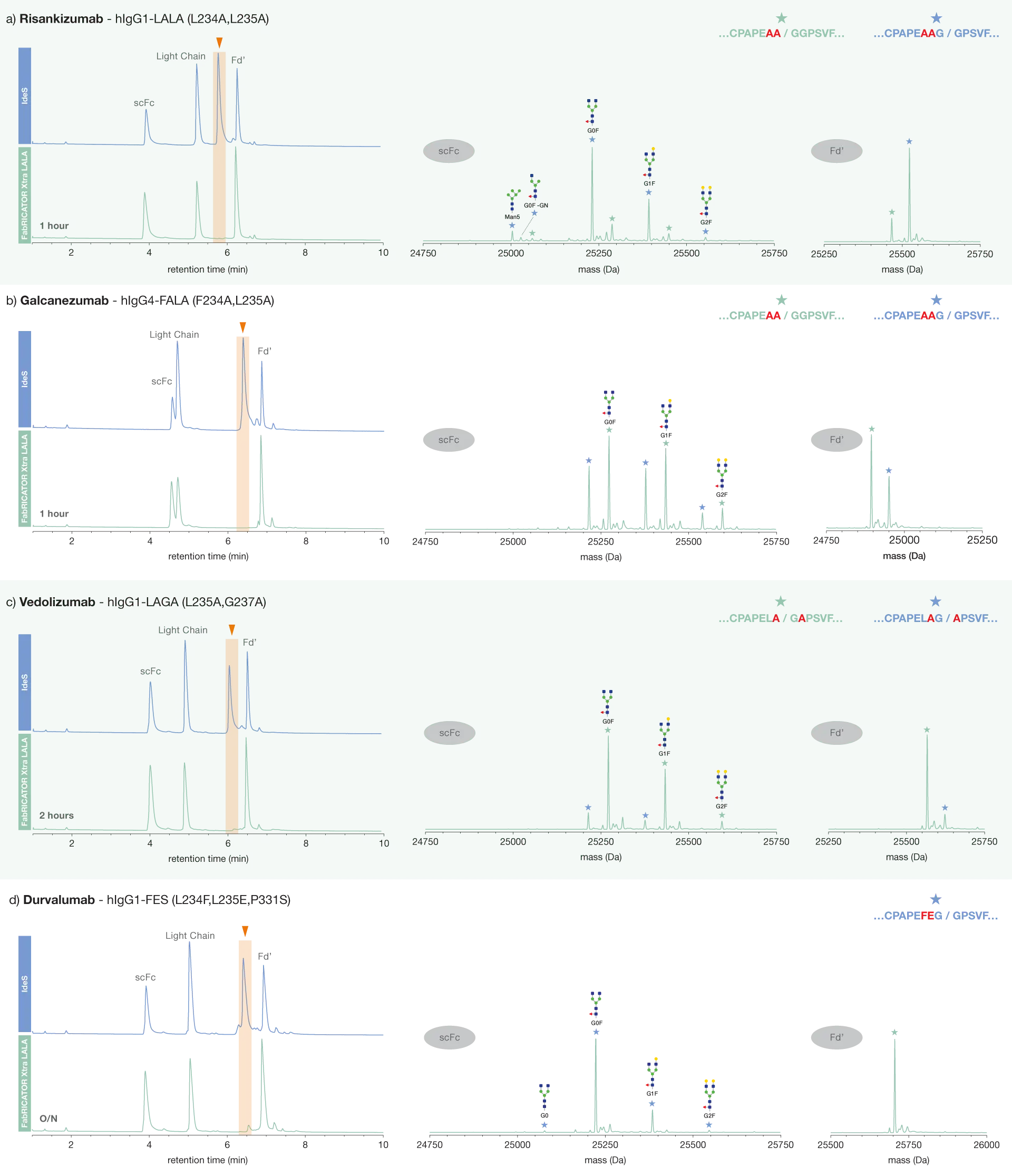
Therapeutic antibodies are frequently engineered to significantly reduce or completely eliminate their Fc effector functions while maintaining their ability to bind antigens. This process, known as Fc silencing, is typically achieved by introducing mutations in the antibody’s hinge region and is especially important in treatments where immune activation could cause harmful side effects. One of the earliest and most widely used strategies is the LALA mutation(L234A, L235A). However, engineering Fc silencing is not a one-size-fits-all solution, and over the years, numerous other mutation combinations have been developed to optimize the therapeutic profile of antibodies.
As most of the mutations in hinge-mutated antibodies, including LALA, occur in the lower hinge region, they can be challenging for IdeS to digest, preventing subsequent subunit analysis. FabRICATOR Xtra LALA digests IgG formats which have been engineered to contain mutated hinge regions below the hinge, generating F(ab’)2 and Fc fragments. These fragments can be reduced to Fd’, LC and scFc for comprehensive subunit analysis of hinge-mutated IgG.
Middle-level characterization with below-hinge antibody digestion
Here, we demonstrate the activity of FabRICATOR Xtra LALA on a variety of engineered antibodies with different combinations of silencing mutations. We tested a molecule containing the classical LALA mutation, a human IgG4 containing the analogous FALA (F234A, L235A) mutation, as well as a human IgG1-LAGA (L235A, G237A) and a human IgG1-FES, which, in addition to the lower hinge mutations L234F and L235E, also contains an additional mutation in the CH2 (P331S).
Digestion condition optimization required for some mutated formats
FabRICATOR Xtra LALA digests all of these formats more effectively than IdeS, allowing for their characterization by LC-MS at the subunit level. In the majority of cases, two FabRICATOR Xtra LALA digestion sites were detected, with digestion occurring after either residue 235 or residue 236.
Improved digestion efficiency of hinge-mutated IgG
Lyophilized enzyme for below hinge digestion of hinge-mutated IgG, including LALA

Download Scientific Poster
