Study on Glycoform Heterogeneity using Enzymatic Digestion and Native Mass Spectrometry
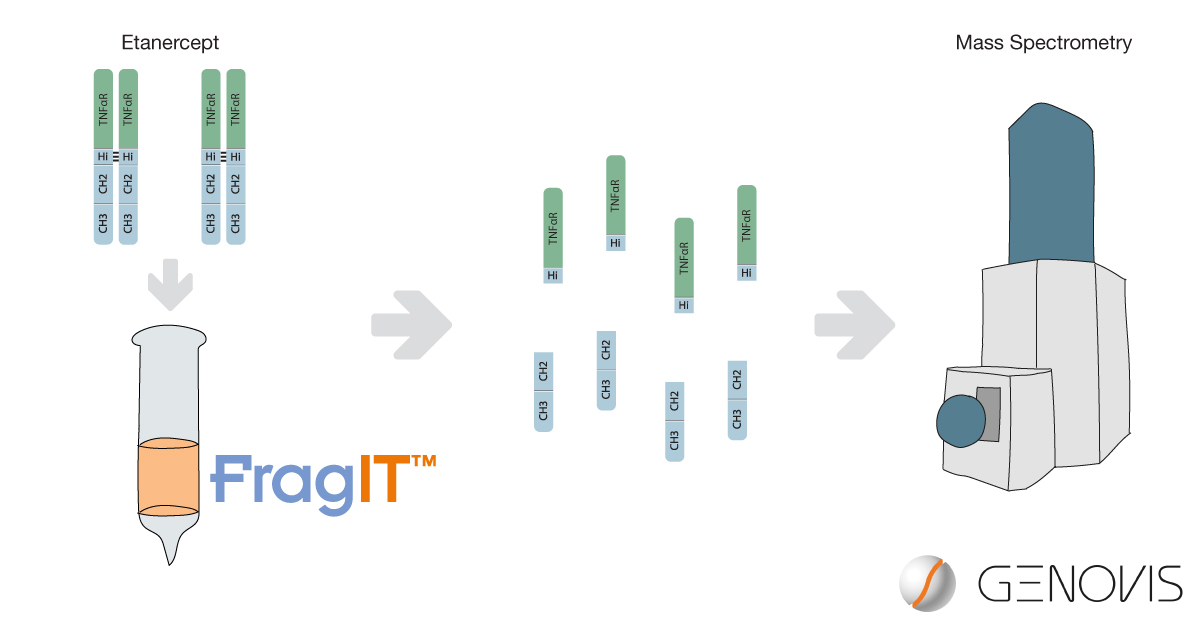
In a study by Wohlschlager et al. (2018), FragIT™ kit was used to digest the Fc-fusion protein etanercept, and the resulting fragments were analyzed using high-resolution native mass spectrometry (MS). Native MS offers a higher spatial resolution at a lower charge state, enabling studies of glycan heterogeneity, and FragIT digestion reduces sample complexity, enabling a detailed annotation of glycoforms on complex compounds.
A detailed knowledge about structure and post-translational modifications (PTMs) is required for biopharmaceuticals to be approved for clinical use, and an important quality attribute that may affect both the efficacy and safety of biopharmaceuticals is glycosylation.
Etanercept is a highly glycosylated Fc-fusion protein that is used to treat autoimmune diseases such as rheumatoid arthritis, and it consists of the TNF-? receptor domain fused to the Fc domain of human IgG1. FragIT – an immobilized version of the FabRICATOR® (IdeS) enzyme – digests IgG from several species and subclasses at a specific site below the hinge region. The resulting fragments are easily purified using the Fc-specific affinity resin that, together with FragIT, comprises the FragIT kit.
In this study, the researchers analyzed the glycosylation of etanercept on both the intact level, and on the middle-down level after FragIT digestion. By combining native MS analysis with enzymatic remodelling of etanercept, a detailed annotation of glycoforms could be achieved and transferred from subunit to whole protein level.
The authors end by concluding:
“Comprehensive information on glycoform heterogeneity, fast analysis with minimal sample preparation and product-characteristic fingerprints render our method highly attractive for the quality control of biologics as well as for comparability studies following changes in the manufacturing process.”(Wohlschalger et al. 2018).
Read more about FragIT and FabRICATOR
References:
