GlycOCATCH™ Affinity Purification
Enrichment resin for affinity purification of mucin-type O-glycoproteins and peptides

Application
Selective enrichment of IgA O-glycopeptides for improved detection and characterization of hinge-region glycosylation.

IgA O-glycopeptides contain heterogeneous O-linked glycans that contribute to IgA structure and function, particularly in the hinge region. Enriching them from an IgA peptide map enhances detection and enables clearer site-specific glycosylation analysis.
IgA antibodies contain both N-linked glycosylation and a heavily O-glycosylated hinge region. To aid understaning of the O-glycosylation within IgA, it is benefical to be able to enrich these portions of the IgA from the rest of the molecule. After trypsin digestion of IgA, the mix of peptides, along with SialEXO, was loaded onto the GlycOCATCH resin before washing with PBS. Elution with 8 M urea demonstrated an enrichment of the expected O-glycosylated hinge tryptic peptide, and various degrees of glycosylation could be identified.
Specific enrichment of IgA O-glycopeptides highlights the presence of multiple O-glycan species
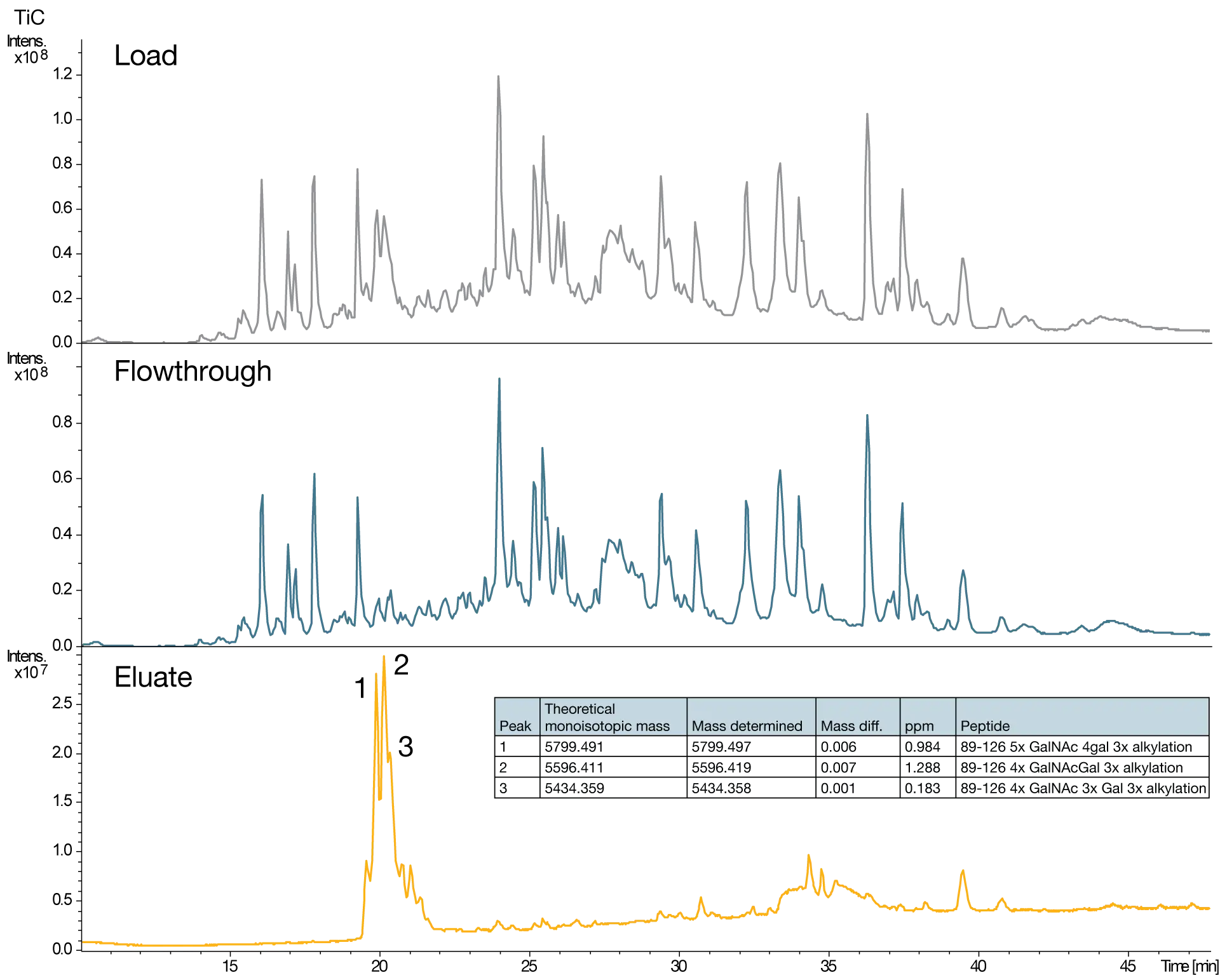
Enrichment resin for affinity purification of mucin-type O-glycoproteins and peptides

Download Product Folder

Download Scientific Poster
