In-depth Analysis of Fusion Proteins with Flexible Linkers
Application
GlySERIAS Immobilized delivers a more complete digestion of linkers for precise characterization of fusion protein quality attributes.

Detailed analysis and identification of quality attributes are of high importance during quality control of protein therapeutics. For fusion protein therapeutics with flexible linkers, this includes confirming the quality of the linked proteins as well as that of the protein linkers. Linker digestion using GlySERIAS may aid in this quality control by separating the linked protein domains. However, the multitude of glycine residues in the linker offers many potential digestion sites of the enzyme, leading to simultaneous hydrolysis at several sites within the linker. The digested product often consists of several variants of the linked proteins with different degrees of the linker remaining. While the observed heterogeneity generally does not negatively impact detailed characterization of the protein subunits, but rather facilitate direct characterization of the linker, a more homogenous digestion product is sometimes required for detailed analysis of the individual protein domains.
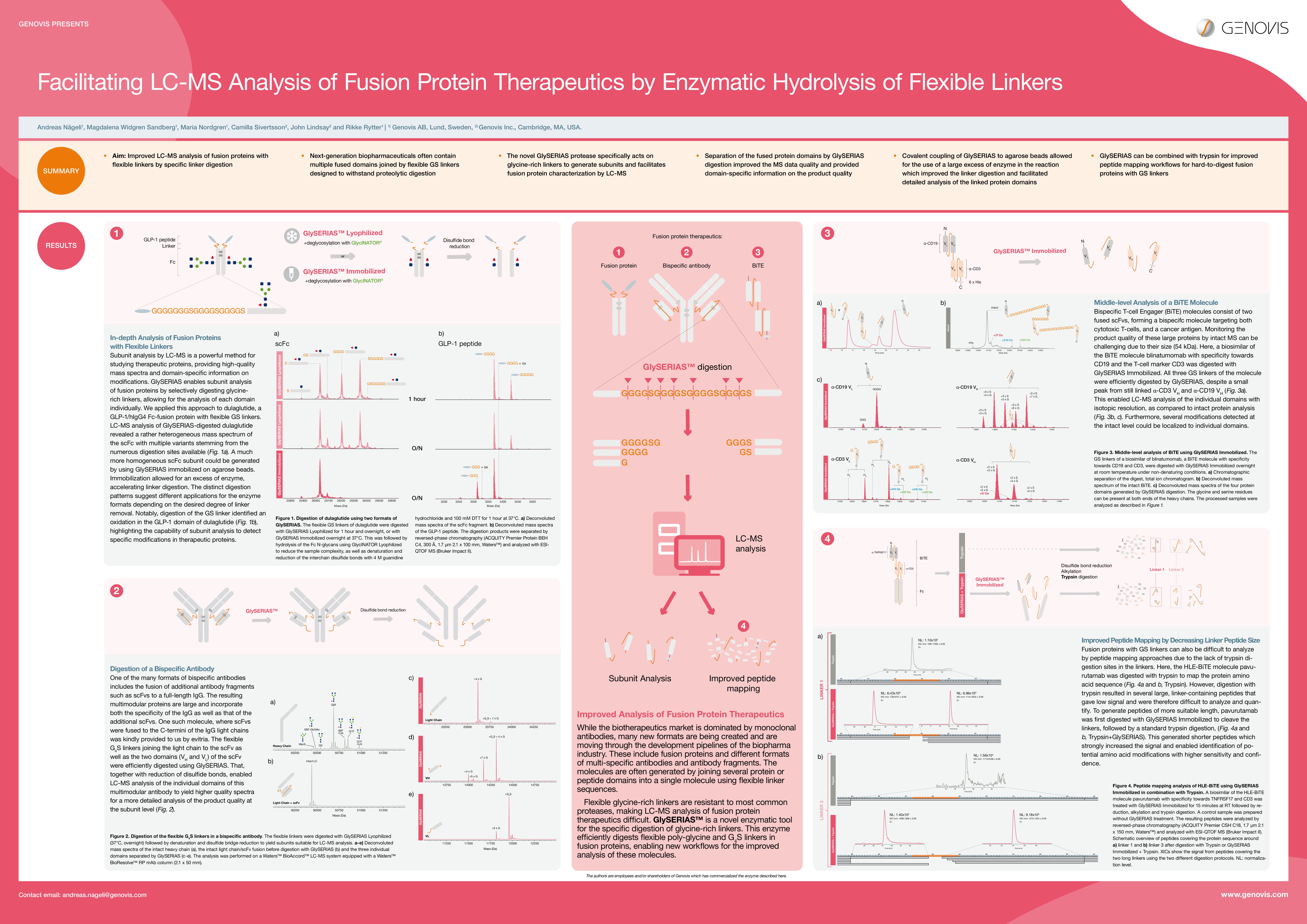
To obtain a more homogenous digestion product of the dulaglutide fusion protein, the protein was digested with GlySERIAS Immobilized, consisting of the GlySERIAS enzyme covalently coupled to agarose beads. By coupling the enzyme to a resin, the enzyme-to-protein ratio can be increased, and thereby the reaction can be accelerated further to a more complete linker digestion. In addition, the enzyme will not be present in the final sample preparation, potentially interfering with sample analysis. For comparison, dulaglutide was digested with GlySERIAS Immobilized overnight at 37°C, or with GlySERIAS Lyophilized for 1 hour and overnight at 37°C. To reduce the sample complexity, the Fc glycans were removed using the endoglycosidase GlycINATOR Lyophilized, and the interchain disulfide bridges were reduced (Fig. 1a).
Analysis of the samples by reversed-phase LC-MS showed that GlySERIAS Immobilized digested the linkers almost completely from the Fc domain, leaving a close to homogenous Fc/2 product containing one serine residue from the linker (Fig. 1b). Digestion with the GlySERIAS Lyophilized for 1 hour or overnight both resulted in several Fc products with different numbers of serine and glycine residues attached. The GLP-1 peptide was present as two variants in all three samples (Fig. 1c). The homogenous Fc/2 from digestion with GlySERIAS Immobilized enabled the detailed study of quality attributes located specifically to the Fc region. Additionally, no enzyme peaks interfering with the analysis were observed in the sample. As a complement, the Fc/2 products with part of the linker remaining from digestion with GlySERIAS Lyophilized, facilitated the study of modifications located to the GS linker.
GlySERIAS Immobilized improves linker removal
Improved linker removal simplifies analysis of the protein domains
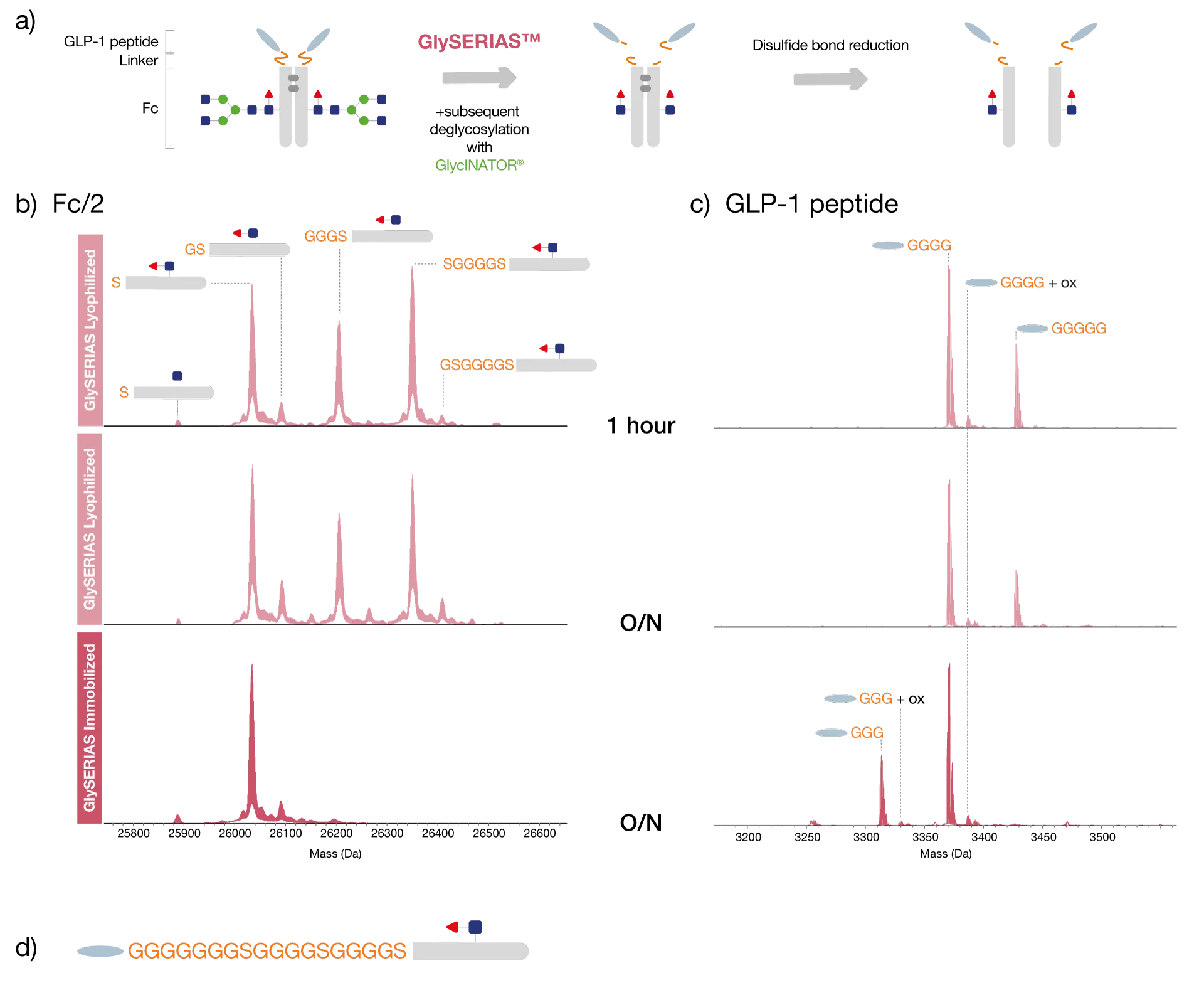
Romiplostim consists of four identical thrombopoietin (TPO) receptor binding peptides and one Fc fragment, linked together by flexible polyglycine sequences. The protein was, similarly to dulaglutide, digested with GlySERIAS Immobilized overnight at 37°C, or with GlySERIAS Lyophilized for 1 hour and overnight at 37°C. The interchain disulfide bridges were reduced to facilitate the following analysis by reversed-phase LC-MS (Fig. 2a).
Similar to what was observed with dulaglutide, digestion with the immobilized enzyme yielded a homogenous Fc/2 with solely one linker residue remaining, here, a glycine residue (Fig. 2b). This facilitated the identification of modifications located specifically in the Fc region. Digestion with the lyophilized enzyme for 1 hour yielded Fc/2 with four remaining glycine residues and a smaller amount of Fc/2 still linked to one TPO peptide. This, on the other hand, enabled the study of the linker region between the Fc/2 and the first peptide without the interference from the second peptide.
GlySERIAS Lyophilized enables linker characterization
Overnight digestion with GlySERIAS Lyophilized yielded two Fc/2 variants with four and one glycine residue remaining, respectively. The peptides released using the lyophilized product were present as five or seven variants, depending on the digestion time, with different numbers of glycine residues remaining from the linker, while the peptides from using the immobilized product were present as four variants (Fig. 2c).
Related Products
Resources
GlySERIAS® for LC-MS Analysis of Fusion Proteins with Flexible Linkers
Download Scientific Poster

AppNote GlySERIAS® for Digestion of Flexible Linkers of Fusion Proteins
Download Application Notes