OmniGLYZOR® Kit
A mix of immobilized enzymes in spin columns for deglycosylation of glycoproteins carrying N- and simple O-glycans

Application
Native removal of O-glycans and removal of inaccessible N-glycans under denaturing conditions, enables complete glycan analysis of the complex C1-inhibitor

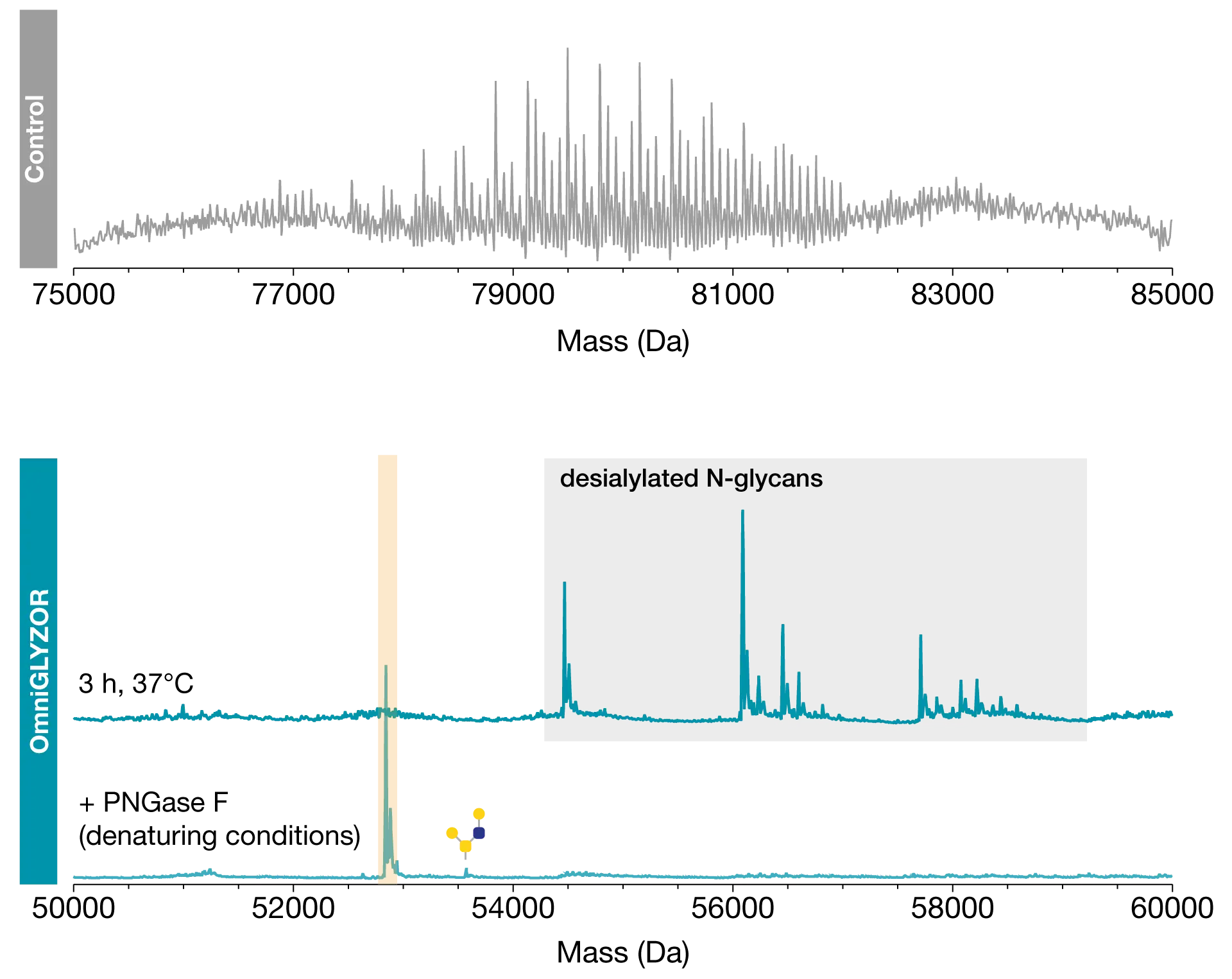
O-linked glycans are generally found at exposed locations on the protein as they are attached after protein folding has occurred. Only such exposed sites are accessible to the glycosyltransferases responsible for their synthesis. N-glycans, on the other hand, are attached co-translationally to the unfolded protein in the ER. This results in certain N-glycosylation sites being poorly accessible to PNGase F or not accessible at all once the substrate protein is folded into its native state. Hydrolysis of such N-glycans therefore requires the substrate protein to be denatured in a way that is compatible with enzymatic activity.
Here we analyzed the C1-inhibitor – a human plasma-derived biotherapeutic modified with 6 N-glycans and up to 28 O-glycans consisting predominantly of sialyl core 1 structures. Without any pretreatment of this highly heterogeneous protein, reversed-phase LC-MS analysis yielded a complex mass spectrum impossible to interpret in detail. Using OmniGLYZOR Microspin columns under native conditions, all O-glycans were efficiently removed within 3 hours. However, between 1 and 3 N-glycans remained on the protein. These inaccessible N-glycans were removed by an additional deglycosylation step under denaturing conditions using the lyophilized PNGase F and the MS-friendly RapiGest™ SF surfactant* included in the OmniGLYZOR kit. Complete removal of all glycans was observed, with the exception of the minor amount of core 2 O-glycans present on the molecule. The enzymes hydrolyzing the O-glycans do not benefit from denaturation, which is why the OmniGLYZOR Microspin columns should only be used under native conditions.
Native deglycosylation of N- and mucin-type-O-glycans
Additional denaturing digestion enables removal of inaccessible N-glycans
Enables identification of low-abundant variants
* RapiGest™ SF Surfactant from Waters Corporation is included in OmniGLYZOR. RapiGest™ is a trademark of Waters Corporation.
A mix of immobilized enzymes in spin columns for deglycosylation of glycoproteins carrying N- and simple O-glycans
