OmniGLYZOR® Kit
A mix of immobilized enzymes in spin columns for deglycosylation of glycoproteins carrying N- and simple O-glycans

Application
Removal of N- and O-glycans from complex and heavily glycosylated proteins, enables clear, interpretable LC-MS analysis.

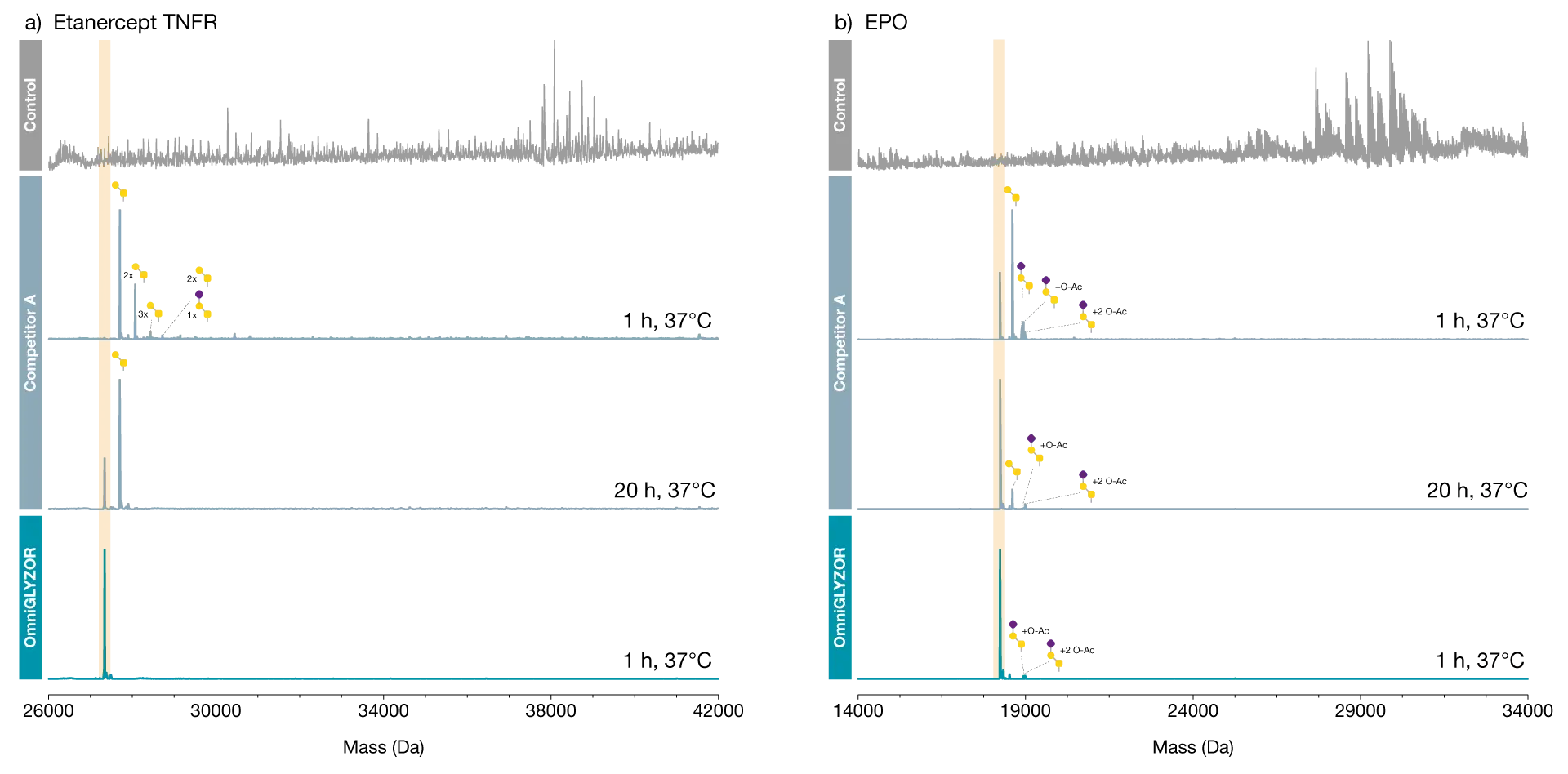
Here we demonstrate the performance of OmniGLYZOR using two therapeutic proteins as substrates. Etanercept is an Fc-fusion protein containing the extracellular domain of the TNFα receptor. This domain is modified with 2 N-glycans and 8-11 core 1 O-glycans with varying degrees of sialylation.
Incubation of this heavily glycosylated protein on OmniGLYZOR Microspin columns for 1 hour at 37°C led to the complete removal of all N- and O-glycans as demonstrated by middle-level LC-MS analysis. Erythropoietin (EPO) is a small protein carrying 3 N-glycans and 1 core 1 O-glycan resulting in close to 40% of its total mass consists of glycans. When analyzed in its intact state, the glycan heterogeneity of EPO results in a very complex mass spectrum.
Efficient deglycosylation of N- and O-linked glycoproteins in a single stepRemoval of glycan heterogeneity simplifies analysis of the protein component
By incubating on OmniGLYZOR Microspin columns for 1 hour at 37°C, the N-and O-glycans are efficiently removed as indicated by a single peak corresponding to the unmodified protein. A minor amount of O-glycans modified with acetylated sialic acids is left on EPO, since such structures are inefficiently hydrolyzed by OmniGLYZOR. Both substrate proteins were also treated with another commercially available deglycosylation product according to the manufacturer’s recommendation (O/N incubation at 37°C) and the data are shown for comparison.
Improves identification of low-abundant variants
A mix of immobilized enzymes in spin columns for deglycosylation of glycoproteins carrying N- and simple O-glycans

Download Product Folder
