High-quality Dynamic Fluorescent Imaging Using GlyCLICK®
Application
Supports high-quality immunofluorescence imaging with consistent signal intensity and reliable quantification through defined antibody labeling.

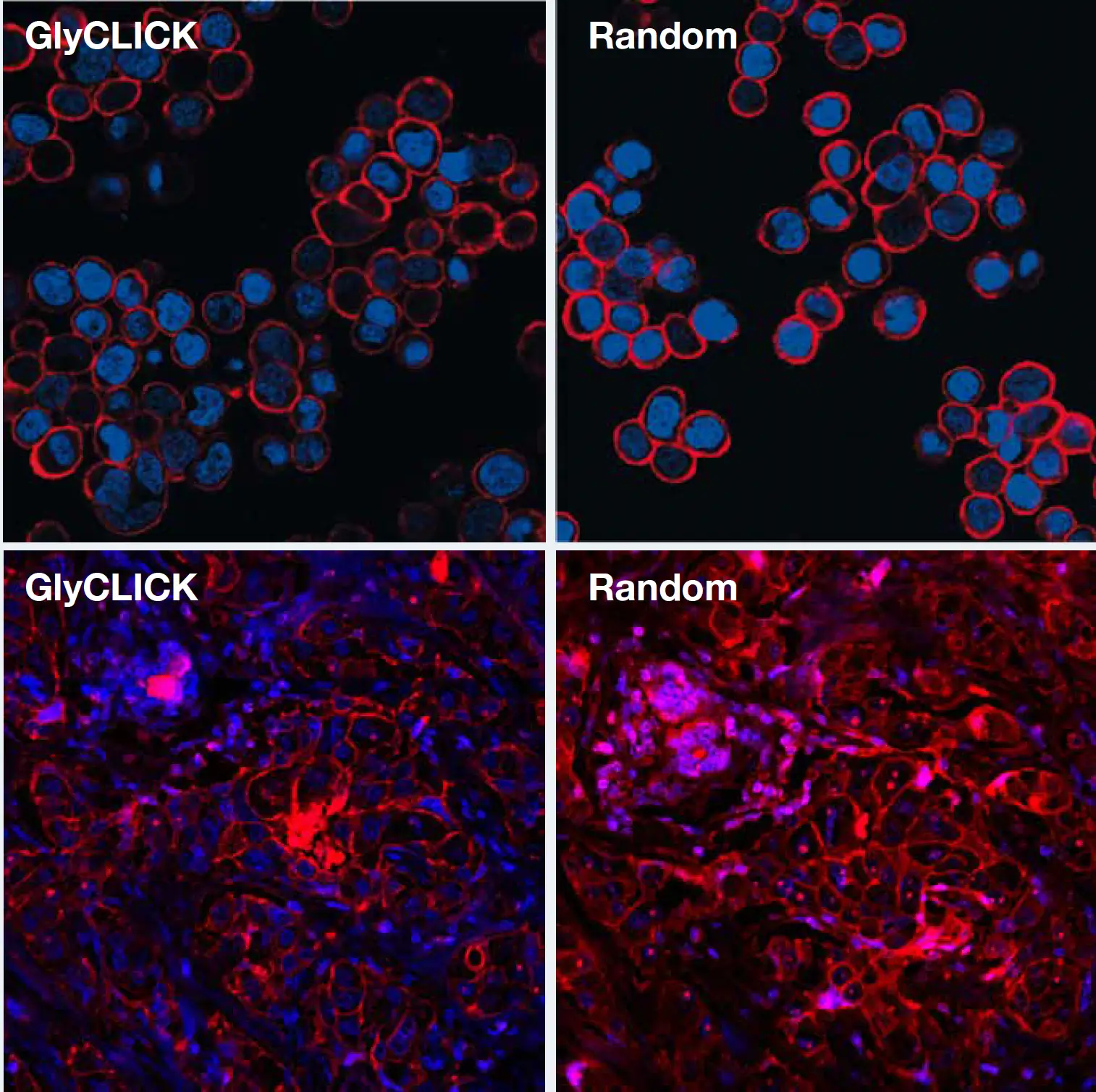
Immunofluorescence microscopy requires not only reliable antigen-binding, but the ability to obtain optimal intensity levels for optical analysis without saturation or loss of signals for high or low antigen expression.
The quality of a microscopy analysis using GlyCLICK conjugated trastuzumab with AlexaFluor®647 was studied by imaging of the HER2 expression on cells and in paraffin-embedded tissue. Imaging of HER2(+) cells and tissue show that T-GlyCLICK-AF647 results in dynamic visualization of antigen distribution and expression levels with minimal noise levels or unspecific staining at optimal intensity levels for minimal saturation or loss of signal.
GlyCLICK conjugation enable dynamic visualization of antigen distribution
The consistent and homogeneous degree of labeling (DOL = 2.0) using GlyCLICK enables more confident quantitation. Generation of effective and reproducible conjugates by random labeling often requires a lot optimization and results in heterogeneous material. In this example, the DOL was in a range of 0-7.
Consistent and homogeneous DOL compared to random conjugation approaches
Related Products
Resources
GlyCLICK®
Download Product Folder

GlyCLICK® for Generation of Site-specific Conjugates for Fluorescent Imaging
Download Scientific Poster

How GlyCLICK® Works
Watch video