Peptide Mapping of Therapeutic Antibodies using GingisREX™
Application
Arginine-specific peptide mapping with improved perfomance over Arg-C, generates larger peptides complementary to trypsin.

Peptide mapping of antibodies using an arginine-specific protease provides a complementary digestion strategy to trypsin by generating an alternative set of peptides, potentially simplifying data analysis and improving sequence coverage.
By digesting only at arginine residues, GingisREX generates larger and fewer peptides than traditional enzymatic tools for peptide mapping, such as trypsin. These larger peptides serve to simplify data-interpretation, as there are fewer peptides to analyse and, by employing complementary digestion approaches to peptide mapping, overall sequence coverage can be improved by combining data sets from complementary digestions.
Complementary digestion approach to trypsin
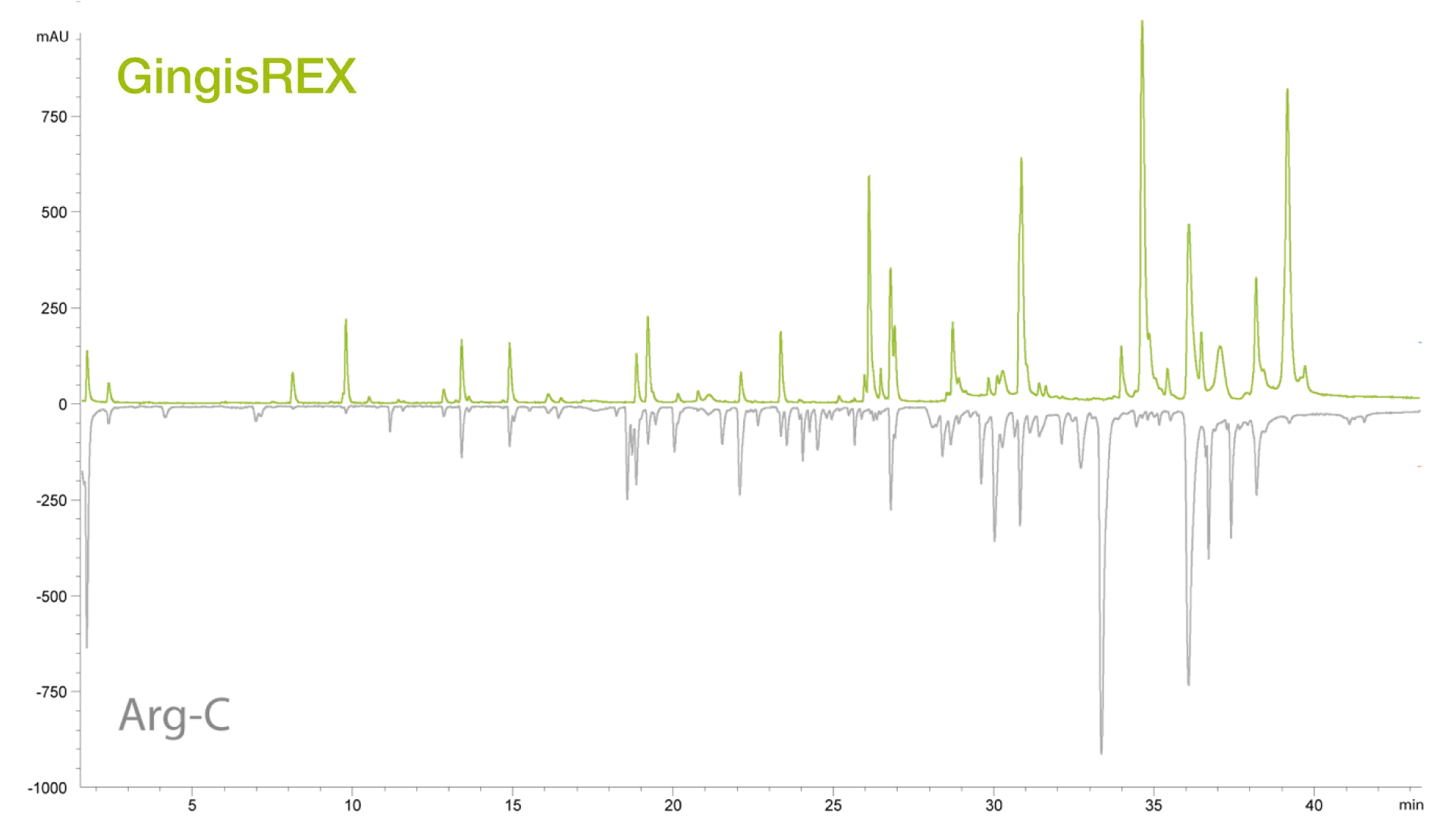
Compared to Arg-C from another source, GingisREX demonstrates a much higher digestion efficiency, generating fewer peptides with lower levels of missed cleavage. The advantage of this increased specificity in peptide mapping applications improves reproducibility, simplifies PTM quantification and increases data confidence. The improved performance of GingisREX, compared with Arg-C, makes this enzyme a much more appropriate choice for performing peptide mapping where this improved level of specificity is required.
Improved specificity to Arg-C increases overall data confidence
Related Products
Resources
GingisREX™
Download Product Folder

GingisREX™ for Specific Digestion at Arginine Residues
Download Scientific Poster




