Reproducible Digestion of a GS-linked Fusion Protein
Robust and reproducible digestion of dulaglutide linkers, separating GLP-1 peptides from the Fc for domain-specific LC-MS analysis of protein modifications

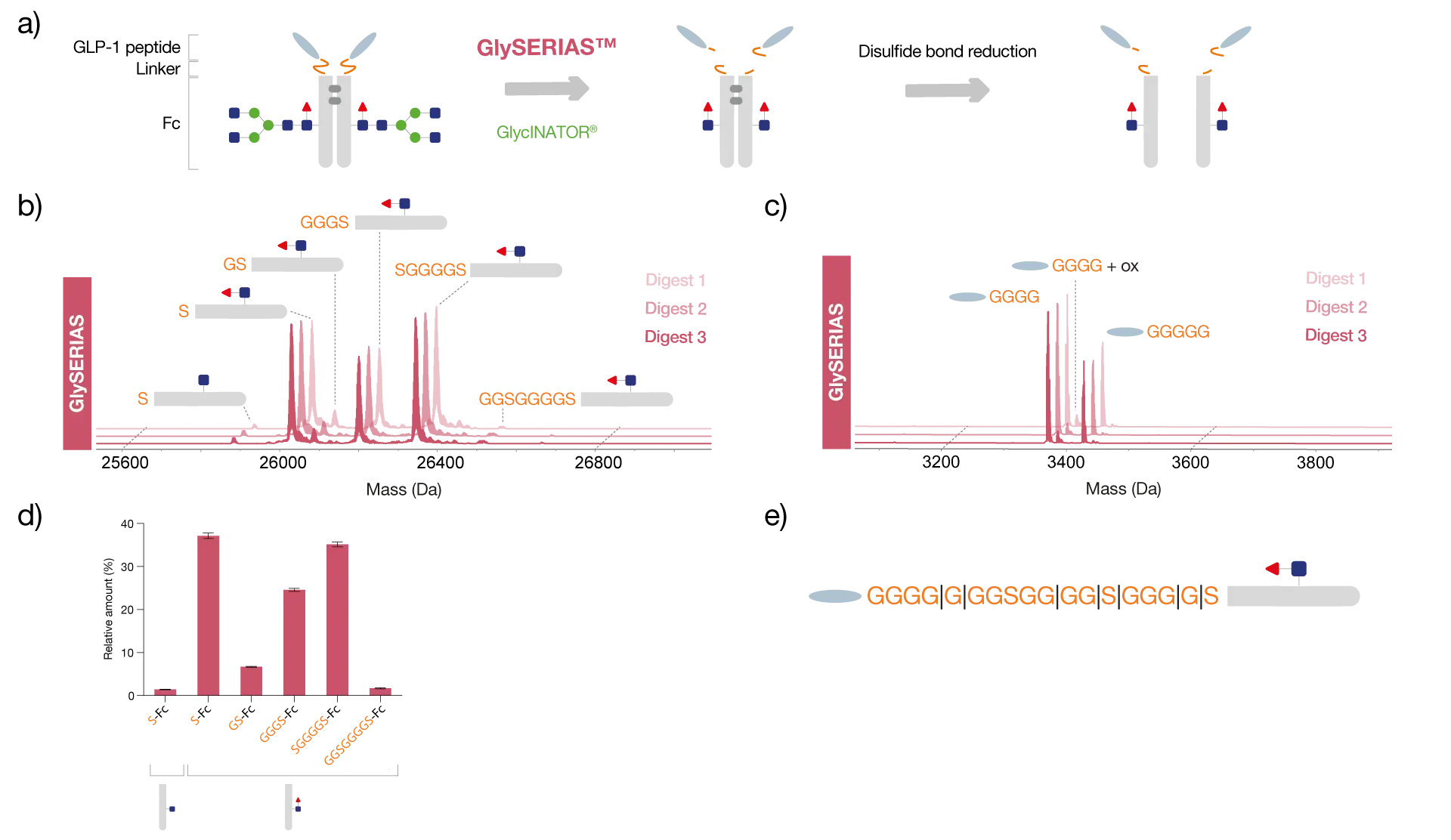
The fusion protein dulaglutide consists of two glucagon-like peptide-1 (GLP-1) molecules linked to the Fc region of human IgG4 via flexible GS linkers.
To study the peptides and Fc region separately, and thereby identify domain-specific PTMs, dulaglutide was digested with GlySERIAS Lyophilized at 37°C for 1 hour. To reduce the sample complexity, the Fc glycans were removed using the endoglycosidase GlycINATOR Lyophilized and the interchain disulfide bridges were reduced with DTT.
Middle-level analysis enables domain-specific localization of modifications
Analysis of the sample by reversed-phase LC-MS showed that the peptides were completely removed from the Fc region upon linker digestion using GlySERIAS. The multitude of glycine residues in the linker offers many different potential digestion sites for GlySERIAS, which is why both the Fc/2 and the GLP-1 peptide were detected as several variants with different numbers of glycine and serine residues still attached.
Efficient digestion of flexible linkers using GlySERIAS
In addition, an oxidation of the GLP-1 peptide was identified. Triplicate digests showed reproducible results in the relative amount of the different Fc/2 variants obtained, despite GlySERIAS digesting at several sites simultaneously.
Reproducible hydrolysis improves confidence in data
Related Products
Resources
GlySERIAS®
Download Product Folder



