Middle-level LC-MS Analysis of Mutated Antibodies

FabDELLO digests human IgG1 at a single site above the hinge, and homogenous Fab and Fc fragments are generated within two hours. The generated fragments can be used for crystallization and NMR studies for higher order structure (HOS), binding studies and much more.

Middle-level LC-MS Analysis of Mutated Antibodies
Mutated hinge regions are common modifications of therapeutic antibody candidates to reduce effector functions. The mutations may affect the antibody digestion efficiency of enzymes with high substrate specificity. For example, the highly specific FabRICATOR (IdeS) digestion efficiency is negatively effected on antibodies containing the common LALA mutation.
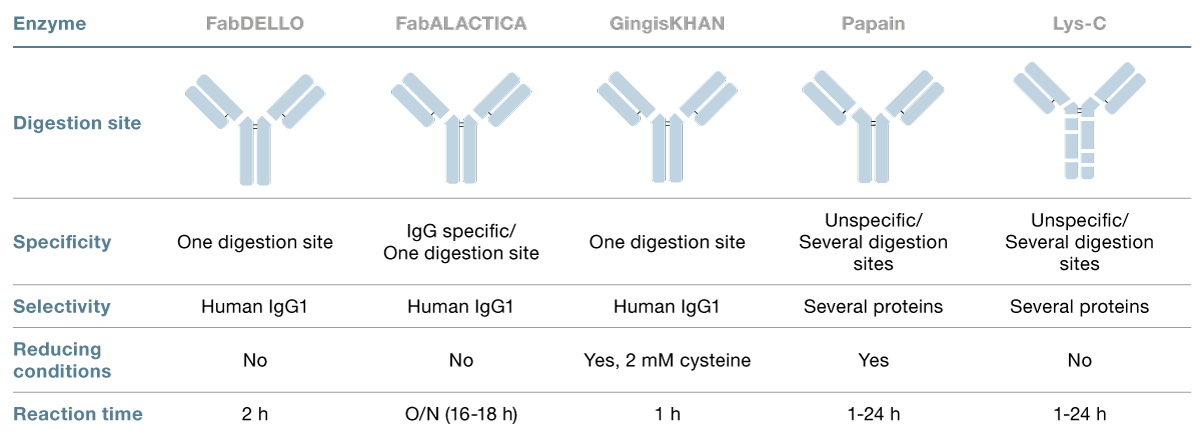
FabDELLO acts at a single exposed lysine residue above the hinge on human IgG1 and enables the popular middle-level LC-MS analysis of antibodies with mutated hinge regions. As an example, the commercially available IgG1 risankizumab, with a LALA mutation in the lower hinge region, was digested with FabDELLO and analyzed by reversed-phase LC-MS. The digestion resulted in generation of homogenous Fab and Fc fragments, as shown in Fig. 1. The fragments were reduced with 20 mM DTT, for middle-level LC-MS analysis, Fig. 2. This generated homogenous Fc/2, light chain (LC) and Fd fragments. The defined peaks observed after digestion with FabDELLO show homogenous fragment generation and robust performance of the enzyme.
Figure 1. The commercial IgG1 risankizumab, with a LALA mutation in the lower hinge, was digested with FabDELLO and analyzed by reversed-phase LC-MS on a Waters™ BioAccord™ system equipped with a Waters™ BioResolve™ RP mAb column (2.1 x 50 mm). Top: UV 280 chromatogram of Fc and Fab subunits analyzed non-reduced. Middle: Deconvoluted Fc spectra. Bottom: Deconvoluted Fab spectra.

Figure 2. The commercial IgG1 risankizumab, with a LALA mutation in the lower hinge, was digested with FabDELLO and the digested material was subjected to 20 mM DTT for 1 h at 37°C to reduce the cysteine bridges. The sample was analyzed by reversed-phase LC-MS on a Waters™ BioAccord™ system equipped with a Waters™ BioResolve™ RP mAb column (2.1 x 50 mm). UV 280 chromatogram showing Fc/2, LC and Fd, and deconvoluted Fc/2, LC and Fd are shown.

Complete Digestion of Mutated Antibodies and Fc-fusion Proteins
Most therapeutic antibodies carry a human IgG1 backbone and the development of bi- and multispecific formats are rapidly increasing. The analysis of such antibodies requires specific enzymes to characterize properties such as monovalent binding, disulfide scrambling and paired Fc glycosylation. Traditional protocols include enzymatic digestion using papain and Lys-C, which require extensive optimizations to minimize overdigestion and obtain sufficient yields.
FabDELLO digests human IgG1 at a single site above the hinge and generates intact Fab and Fc fragments. To demonstrate the specific activity of FabDELLO, a selection of IgG1 and Fc-fusion proteins were digested and analyzed by non-reduced SDS-PAGE. A complete digestion was achieved for all substrates, including IgG1 with the otherwise difficult LALA and LFLE mutations. The precise and effective performance of the FabDELLO enzyme on this wide range of substrates validates its use for the development of biopharmaceuticals.
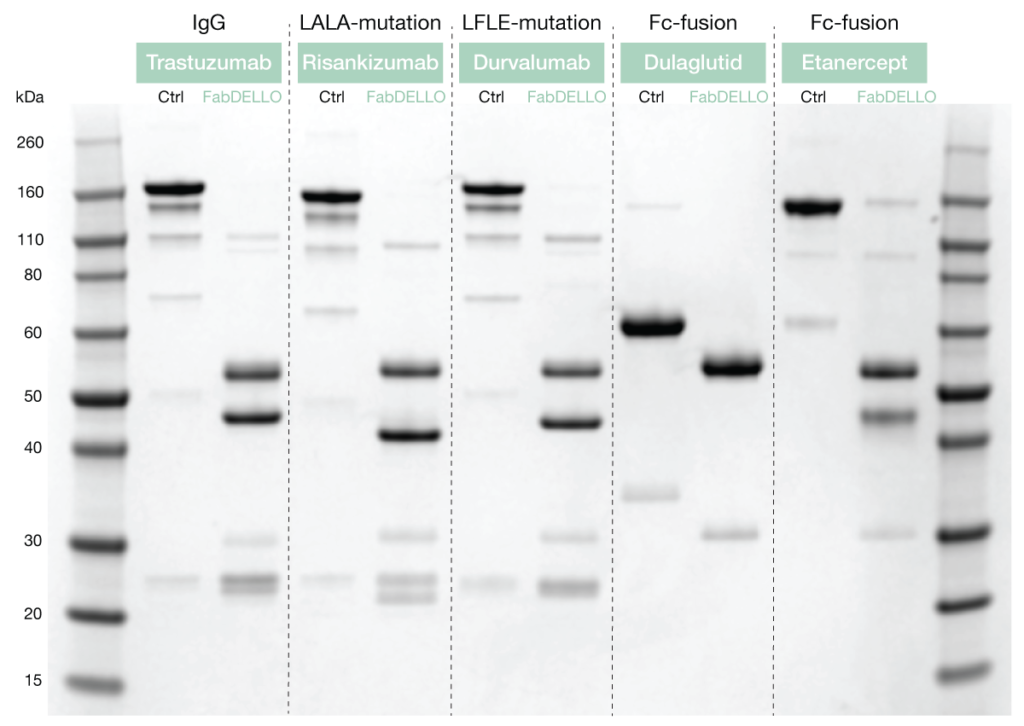
Figure 3. Non-reducing SDS-PAGE analysis of commercial antibodies and Fc-fusion proteins digested with FabDELLO.

Comparison of Enzymatic strategies for Obtaining Fab Fragments
For applications involving intact Fab or Fc fragments, it is necessary to digest the antibody in the hinge region. Traditional enzymes such as papain and Lys-C can be used to generate Fab fragments, however, careful optimization is needed to minimize the risk of unspecific digestion. FabDELLO has a single digestion site at the exposed lysine above the hinge of human IgG1, and homogenous Fab and Fc fragments are generated within two hours. The generated fragments can be used for crystallization and NMR studies for higher order structure (HOS), binding studies and much more.