Articles tagged ”Glycosylation”
FabULOUS™ in HIV Vaccine Research
The Fc N-glycosylation profile can be linked to non-neutralizing functions in a vaccine. In a recent study by Chen et al., FabULOUS digestion provided valuable insights in HIV vaccine research following N-glycosylation analysis of the Fc region in mouse antibodies.
Read more »

A Middle-up Approach using FabALACTICA for Characterization of Bispecific Antibodies
In recent years, bispecifics have gained popularity due to their therapeutic advantages over conventional IgG’s. In particular, the T-cell bi-specifics have received a great deal of attention due to their potential for improved efficacy. However, because of their complex TCB formats, there are multiple challenges associated with manufacturing and analysis of these type of biomolecules. A number of product and process related side products are formed which require close monitoring and identification. Moreover, the existence of various charge variants is common which can be challenging to fully characterize and understand.
Read more »

C-terminal lysine clipping and Fc receptor binding using SmartEnzymes
Researchers at LFB Biotechnologies in Paris, France have carried out a thorough analysis and characterization of the impact of C-terminal lysine clipping to Fc-receptor binding using a range of SmartEnzymes from Genovis.
The scientists separated an IgG1 antibody using SCX separation and purified the fractions without C-terminal lysines K0, with 1 C-terminal lysine K1 and with both lysines intact K2. The purified fractions were characterized for any further differences using FabRICATOR digestion and middle-level analysis. This approached enabled the researchers to study multiple post-translational modifications such as charge variants, oxidations and Fc glycosylation in a simple and robust way. The characterization revealed that the lysine heterogeneity was the main differentiator and all other PTMs were distributed between the fractions.
Read more »

OpeRATOR Publication from Johns Hopkins University
Scientists from the prestigious Johns Hopkins University School of Medicine have used OpeRATOR to develop a workflow to map O-glycosylated sites on proteins in very complex samples. O-glycoproteins are notoriously difficult to study due to the low abundance, high structural heterogeneity and low stability. Previous approaches using affinity enrichment or engineered cell culture systems either lack efficiency or are ill-suited forO-glycoproteomic studies of complex samples.
In the workflow developed by Weiming Yang and colleagues, protein samples such as serum or kidney tissue were digested with trypsin, immobilized onto beads through the N-terminus and treated with OpeRATOR and SialEXO. OpeRATOR is an endoprotease and derived from the gut commensal bacteria Akkermansia muciniphila that specifically cleaves peptides and proteins N-terminally of O-glycosylated serine or threonine residues. Therefore, only O-glycopeptides are released from the solid support and were identified using ETD mass spectrometry.
Using this workflow, Yang et al. were able to map over 3000 O-glycosylation sites from human serum, T cells and kidney tissue, almost doubling the number of known O-glycosylation sites. They were also able to detect and quantify the aberrant O-glycosylation patterns in kidney tumors, showcasing the potential use of such methodologies for both basic research and diagnostic purposes.
Meet the Scientist
We got the opportunity to interview the first author of the paper, Weiming Yang at Johns Hopkins University.


For more information on OpeRATOR go the the following pages:
- OpeRATOR Product Page
- Analysis of O-glycosylated proteins
- Download poster on OpeRATOR from ASMS 2018


Study on Glycoform Heterogeneity using Enzymatic Digestion and Native Mass Spectrometry
In a study by Wohlschlager et al. (2018), FragIT™ kit was used to digest the Fc-fusion protein etanercept, and the resulting fragments were analyzed using high-resolution native mass spectrometry (MS). Native MS offers a higher spatial resolution at a lower charge state, enabling studies of glycan heterogeneity, and FragIT digestion reduces sample complexity, enabling a detailed annotation of glycoforms on complex compounds.
A detailed knowledge about structure and post-translational modifications (PTMs) is required for biopharmaceuticals to be approved for clinical use, and an important quality attribute that may affect both the efficacy and safety of biopharmaceuticals is glycosylation.
Etanercept is a highly glycosylated Fc-fusion protein that is used to treat autoimmune diseases such as rheumatoid arthritis, and it consists of the TNF-? receptor domain fused to the Fc domain of human IgG1. FragIT – an immobilized version of the FabRICATOR® (IdeS) enzyme – digests IgG from several species and subclasses at a specific site below the hinge region. The resulting fragments are easily purified using the Fc-specific affinity resin that, together with FragIT, comprises the FragIT kit.
In this study, the researchers analyzed the glycosylation of etanercept on both the intact level, and on the middle-down level after FragIT digestion. By combining native MS analysis with enzymatic remodelling of etanercept, a detailed annotation of glycoforms could be achieved and transferred from subunit to whole protein level.
The authors end by concluding:
“Comprehensive information on glycoform heterogeneity, fast analysis with minimal sample preparation and product-characteristic fingerprints render our method highly attractive for the quality control of biologics as well as for comparability studies following changes in the manufacturing process.”(Wohlschalger et al. 2018).
Read more about FragIT and FabRICATOR
References:
ADC Subunit Characterization of Drug Load and Glycosylation using HILIC-MS
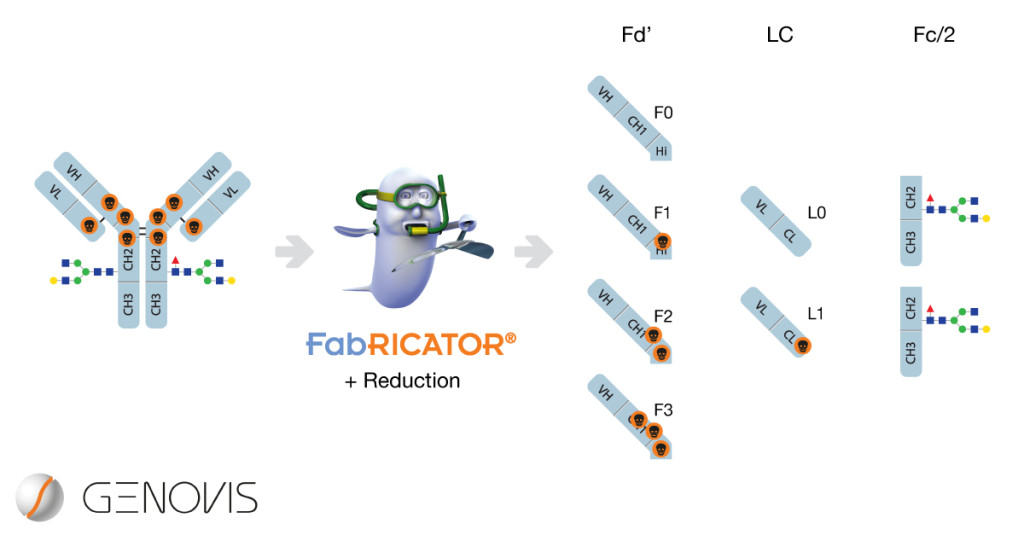
In a collaboration headed by Davy Guillarme at University of Geneva, scientists have explored the characterization of subunits derived from antibody drug conjugates (ADCs) using hydrophilic interaction chromatography (HILIC) coupled to mass spectrometry (D’Atri et al. 2018).
The scientists used brentuximab vedotin (BV, Adcetris®), an approved ADC for treatment of Hodgkin lymphoma (HL) and systemic anaplastic large cell lymphoma (ALCL). The BV consists of an antibody directed towards CD30, coupled to the vedotin toxin using cysteine conjugation chemistry. The random cysteine conjugation method results in a heterogeneous attachment of the drug, with differences in efficacy depending on the drug load. For this reason, the amount of conjugated toxins requires careful characterization. A key quality attribute of both antibodies and ADCs is the glycosylation profile, that may affect the stability, efficacy and safety. In this paper, a method to study ADC drug load and glycan profiling in a single experiment was demonstrated.
The intact ADC is around 150 kDa, which makes it very complicated to study details with high resolution. For this reason, D’Atri and colleagues used FabRICATOR digestion and reduction to generate specific antibody subunits of around 25 kDa, with increased resolution in both separation and mass determination. New wide-pore HILIC phase has enabled separation of larger molecules such as antibody subunits, and the team has already published a glycoprofiling strategy using HILIC on naked antibodies (Periat et al. 2016).
The coupling of HILIC separation to MS of subunits resulted in more detailed characterization of the subunits as compared to reverse phase separation (RP-HPLC). The relative percentage of each subunit aligned well with both methods of separation. However, additional positional isomers of the Fd’ fragment were observed using HILIC separation. Also, the glycoforms of the Fc/2 fragments were chromatographically separated, making mass deconvolution and determination easier. The authors conclude the middle-up HILIC-MS method to be orthogonal to RP-MS with the benefit that the methodology allows simultaneous characterization of drug load and glycosylation of the antibody drug conjugate.
FabRICATOR is a protease with a single digestion site below the hinge of IgG. The enzyme is widely used in middle-level analytical workflows for characterization of antibody based biopharmaceuticals. Learn more about FabRICATOR.
References
Periat, A. et al., 2016. Potential of hydrophilic interaction chromatography for the analytical characterization of protein biopharmaceuticals. Journal of chromatography. A, 1448, pp.81–92.

