
Localization of O-glycan Sites in CD24Fc by OpeRATOR® Digestion and LC-MS Analysis
Scientists from Merck & Co. and Glycotope describe the localization of 12 O-glycosylation sites in the heavily O-glycosylated fusion protein CD24Fc by digestion with the O-glycoprotease OpeRATOR in combination with LC-MS analysis. Two different workflows were developed, one based on fractionation in combination with LC-MS/MS CID and one based on LC-MS/MS EThcD. OpeRATOR digestion enabled the localization of O-glycan sites in both workflows and relative quantification of different glycoforms.
Human cluster of differentiation (CD24) is a highly glycosylated peptide which upon binding to its receptor sialic acid-binding Ig-like lectin 10 (Singlec-10), expressed on the surface of immune cells, induces suppression of the immune response. Fusion of CD24 with an Fc domain (CD24Fc) increases the serum half-life of CD24 and has been demonstrated to decrease chronic immune response activation and inflammation to control the progression to AIDS (acquired immunodeficiency syndrome).
CD24 consists of approx. 30 amino acids with 16 potential O-glycosylation sites and two N-glycosylation sites. The glycosylation, and especially sialic acids, is critical for the interaction between CD24 and Singlec-10. Detailed characterization of the O- and N-glycosylation of CD24 contributes to the understanding of the different roles the glycosylation play in the molecule. While previous studies have characterized the protein N-glycosylation in a quantitative and site-specific manner, the excessive protein O-glycosylation have only been studied from released glycans because of the lack of appropriate tools to achieve site-specific information about the protein O-glycosylation.
In this study, digestion of CD24Fc using the O-glycoprotease OpeRATOR enabled mapping of the O-glycans sites and provided quantitative data on the O-glycan composition of CD24. OpeRATOR is an O-glycan-specific protease that digests proteins carrying core 1 O-glycans, N-terminally of the glycosylated serine or threonine residues.
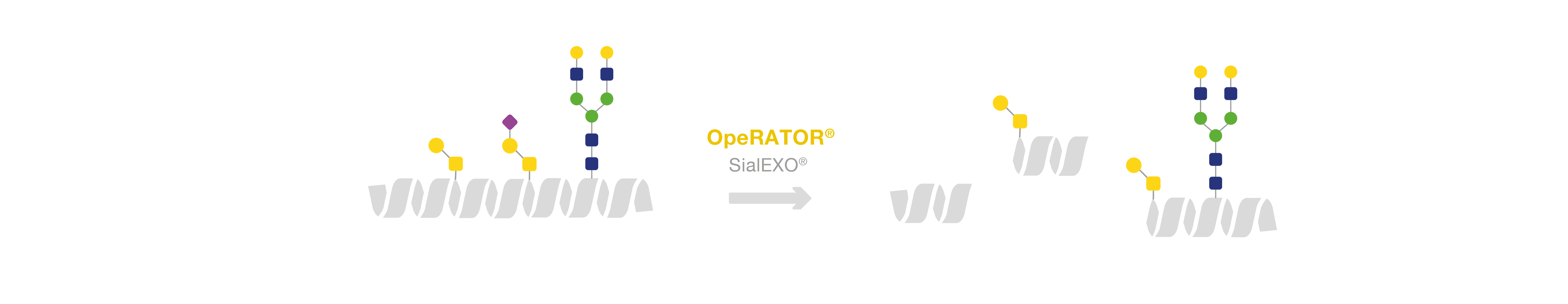
Two different workflows were developed based on LC-MS analysis and fragmentation through either collision-induced dissociation (CID) or electron-transfer/higher-energy collision dissociation (EThcD). In the first workflow, O-glycopeptides were generated by digestion of CD24Fc using FabRICATOR, PNGase F and trypsin/Asp-N. The O-glycan sites and structures of the peptides from the Asp-N digestion were determined by fractionation of the peptides, followed by digestion using OpeRATOR and SialEXO and analysis by LC-MS/MS CID. This revealed seven O-glycan sites in CD24 and gave a relative quantification of the structures and sites. Since the fragmentation by CID does not retain the glycans on the fragment ion, glycan site determination of heavily glycosylated peptides by CID is not always feasible. Therefore, a second workflow was developed based on fragmentation by EThcD. Here, O-glycopeptides were directly generated by digestion using OpeRATOR and SialEXO followed by LC-MS/MS EThcD analysis. Limited Lys-C digestion was used to remove the Fc domain from the most C-terminal O-glycosylated peptide and PNGase F was used to remove the N-glycans. This strategy enabled identification of another five O-glycosylation sites, besides the already identified seven sites.
Altogether, OpeRATOR digestion enabled identification of a total of 12 O-glycosylation sites in CD24. T25 was identified as the predominant O-glycosylation site and NeuAc(1)Hex(1)HexNAc(1) was identified as the major O-glycoform. The workflow based on fractionation and CID was most favourable for identification of singly O-glycosylated peptides and provided relative quantification information, while the workflow based on EThcD was most useful for identification of singly, doubly and triply O-glycosylated peptides.
The data presented in this study illustrate the benefit of employing an O-glycoprotease like OpeRATOR in combination with LC-MS to identify the O-glycan sites in heavily O-glycosylated proteins. It also illustrates the strength of combining different sets of enzymatic tools with different LC-MS/MS fragmentation strategies to investigate the O-glycosylation profiles of specific proteins.
Reference
Li et al., 2022. Precise O‑Glycosylation Site Localization of CD24Fc by LC-MS Workflows Anal. Chem. https://doi.org/10.1021/acs.analchem.2c01137.
OpeRATOR® – O-glycan-specific protein digestion

